| Journal of Cancer Stem Cell Research (2014), 2:e1006 © 2015 Creative Commons. All rights reserved ISSN 2329-5872 DOI: 10.14343/JCSCR.2015.3e1002 http://cancerstemcellsresearch.com |
 |
| Journal of Cancer Stem Cell Research (2014), 2:e1006 © 2015 Creative Commons. All rights reserved ISSN 2329-5872 DOI: 10.14343/JCSCR.2015.3e1002 http://cancerstemcellsresearch.com |
 |
| Research Article | Open Access |
| Autophagy reduces subpopulation of CD44+/CD24−/low phenotype cancer stem cells in MCF7 and Hep-2 cells culture | |
| Babak Nami1,*, Huseyin Donmez2 and Nadir Kocak3 | |
| Department of Medical Genetics, Selcuk University Medical Faculty, Konya, Turkey | |
| *Corresponding author: Babak Nami, Department of Medical Genetics, Selcuk University Medical Faculty, Alaaddin Keykubat campus, 42075/Selcuklu, Konya, Turkey. Tel: 0090 332 224 3933; Fax: 0090 332 224 3930; E-mail: nami@selcuk.edu.tr Received: December 11, 2014; Revised: January 28, 2015; Accepted: February 2, 2015 | |
Abstract: Growing evidences indicate that tumor regrowth and invasion refer a small subpopulation of tumor cells called cancer stem cells (CSCs). Autophagy is an essential catabolic process for cell homeostasis that involves in cell survival and death and also development of various human diseases including cancer. Here we aimed to investigate the effect of autphagy on the subpopulation of CD44+/CD24−/low phenotype MCF7 and Hep-2 cells. CD44+/CD24−/low cells were isolated from MCF7 and Hep-2 cell cultures by using fluorescence-activated cell sorting (FACS). The CD44+/CD24−/low and the original cells were separately treated with 20 μM of C2 ceramide for 24 hours in order to stimulation of autophagy. LC3 proteins aggregation was monitored to evaluate autophagy in the cells by using LC3 targeted immunofluorescence staining assay. Subpopulation of CD44+/CD24−/low cells in total cultures were investigated by flow cytometry analysis and eventually statistics were carried out. Results showed an enhanced aggregation of LC3 proteins in the cells and also CD44+/CD24−/low cells under effect of C2 ceramide. Moreover, the subpopulation of CD44+/CD24−/low cells were found significantly lower in the cells cultures treated with C2 ceramide in comparison with negative controls. The results elucidated that CD44+/CD24−/low MCF7 and Hep-2 cells were susceptible to autophagy stimulatory reagent C2 ceramide. Additionally, our results suggest that CD44+/CD24−/low cell subpopulation in the cell cultures is reduced under autophagic condition. Therefore, we suppose that autophagy may has a negative impact on the breast and Larynx cancer stem cells self-renewal potency or expression of CD44 and CD24 cell surface markers, however further studies are needed to reveal the exact mechanism.
Keywords: Autophagy, Cancer, Stem cells, CD44, CD24, MCF7, Hep-2.
Autophagy is a highly conserved catabolic pathway that targets dysfunctional cellular organelles and cytoplasmic constituents that leads to cell degradation and elimination of cytoplasmic components by lysosomal machinery [1]. Autophagy contributes in genomic stability and protecting cells against stress, organelle damage and microbes [2]. In addition, it regulates cellular homeostasis during synthesis and is necessary for removal damaged or long-lived organelles and proteins [1, 2]. Autophagy has been referred to as a double-edged sword because of having a dual role in cell survival and death [3]. The dual role of autophagy in the promotion also prevention of many tumors has been demonstrated previously [3, 4]. Numerous literatures indicated that cells possess a mechanism of programmed cell death that is associated with the formation of autophagosomes and depends on autophagy-depending proteins [5]. This suggests that autophagic programmed cell death is an alternative mechanism to apoptosis [6]. Autophagy assist tumor cells to tolerate stressful metabolic environments qua some types of cancer depend on autophagy to advancement and survival [7, 8]. A growing area of evidence implicates a paradoxical role of autophagy following anticancer treatments as well. Cancer cells can exploit autophagy to resist the cytotoxicity of anticancer drugs, although some drugs may require autophagy to kill cancer cells [9]. In contrast, it can prevent the initiation of tumorigenesis through protecting genomic stability, reducing cytoplasmic damage also inflammation during the initial stage of tumor formation [10]. The lack or inhibition of autophagy leads to increased levels of reactive oxygen species which causes the accumulation of DNA damage. This appears as gene amplification, polyploid nucleus and increased DNA double-strand break relating a perfect target to anticancer therapy [11]. Thereby, this mechanism has been a strong motivating factor for determining cells fate.
Recently, researchers propagate that the capability of a tumor to grow and chemoresistance involved to on a small subset of tumor cells termed cancer stem cells (CSCs). These cells are believed to be responsible for tumor initiation, recurrence and failure of conventional therapies. [12]. Cancer stem cells are defined as undifferentiated cells, having stem cell properties with self-renewal potency and ability to regenerate malignant cells and act as an engine to growth of the cancer [13]. According to many studies, invasion, regrowth potency and treatment of cancer is connected to function of CSCs. The CSCs have been described as possessing the cell-surface phenotype CD44+/CD24−/low cells that represent averagely 1–2% of all cells in a tumor [14] and enhancement in population exhibits increased tumorigenicity and invasion [15, 16]. CD44 is a cell-adhesion molecule responsible in binding of cells to hyaluronic acid [17], whereas CD24 is a regulator of CXCR4, a molecule involved in breast tumor formation [18]. It has been revealed that CD44+/CD24−/low subpopulation of primary tumors has a direct linkage with resistance against chemotherapies [19], metastasis and malignant relapse [20]. Although data have been provided to support resistance of CSCs against programmed cell death mechanisms [21], researchers believe CSCs could be Achilles' heel of cancer in CSC targeting therapies [12]. In this study we aimed to investigate MCF7 and Hep-2 derived CD44+/CD24−/low cell subpopulation in a culture model under autophagic condition.
C2 ceramide was purchased from Sigma-Aldrich (St. Louis, USA) and were dissolved in dimethyl sulfoxide (DMSO) as 10 mg/ml and stored at a +4°C until use. Rabbit anti human MAP LC3 IgG, rabbit anti human MAP LC3 IgG and FITC-conjugated goat anti-rabbit IgG antibodies were purchased from Santa Cruz Biotechnology Inc. (Dallas, Texas, USA). FITC-conjugated mouse anti human CD44 monoclonal and PE-conjugated mouse anti human CD24 monoclonal antibodies were purchased from Thermo scientific Inc (Rockford, IL, USA).
MCF7 and Hep-1cell lines were purchased from the American Type Culture Collection (ATCC). The cells were cultured in Dulbecco's Modified Eagle's Medium (DMEM; Gibco) that was completed with 10% fetal bovine serum (Gibco), 1% L-glutamine (Gibco), 1% penicillin (100 unit/ml) and 1% streptomycin (10 mg/ml) with incubation at 37°C in a 5% CO2 humidified incubator. After the cell culture reached 80% confluency, cells were trypsinized with 0.25% trypsin (Sigma-Aldrich, St. Louis, USA) and then harvested. In order to perform the experiments, the cells were seeded as 2 × 104, 5 × 104, 105 and 5 × 105 cells in 96, 24, 12 and 6-well plates respectively in triplicate for each one and then cultured at the condition maintained above for 24 hours before start to treatment.
Cells were cultured on immunofluorescence slides 24 hours before treatment. At the end of the treatment period, the slides were rinsed in PBS and the cells were fixed by cold methanol for 5 minutes. Blocking was done with incubation of slides in 1% BSA solution for an hour with gentle agitation. After the blocking, the slides were incubated in 1 μg/ml solution of rabbit anti MAP LC3 IgG as the primary antibody for an hour with gentle agitation. Then, the slides were rinsed in TBST tree times each for 5 minutes. Afterwards the slides were incubated in 1 μg/ml solution of goat anti-rabbit IgG-FITC as the secondary antibody for an hour in darkness. The slides were then washed completely in TBST and were mounted by 4′,6-diamidino-2-phenylindole (DAPI). The slides were observed under a confocal fluorescence microscope (Nikon, Japan).
Cells were trypsinized and washed with HBSS and were pelleted by spinning down. One million cells were resuspended in 100 μl of 2% FBS/HBSS solution and then were transferred to a test tube. 20 μl from each FITC-conjugated mouse anti human CD44 monoclonal and PE-conjugated mouse anti human CD24 monoclonal antibodies (Thermo scientific, Rockford, USA) were added to the tube and then incubated at +4°C in darkness for an hour with gentle agitation. Afterwards, the cells were rinsed two times with 2% FBS/HBSS solution and 400 μl of 1 μg/ml DAPI solution (dissolved in 2% FBS/HBSS) were added to the cells. The cells were analyzed using FACS machine (BD FACSAria™ III cell sorter) with adjusting same gape for each capillary in terms of CD44 and CD24 markers expression rate. FITC-and PE- conjugated isotype control antibodies (Thermo Scientific, Rockford, USA) were used as positive control and unstained cells were as negative control. The subpopulations were calculated among living cells.
The statistical significances were considered applying two-tailed student's t test and analysis of variance (ANOVA) using the GraphPad Prism® V.5.00 software (La Jolla, San Diego, USA). The image base data were analyzed using ImageJ software (NIH, Maryland, USA). P < 0.05 was considered as statistically significant.
In order to confirm susceptibility of breast CSCs against autophagy, we monitored the LC3 protein aggresomes in the FACS sorted CD44+/CD24−/low cells treated with 20 μM C2 ceramide for 24 hours in serum free medium. The result showed an enhanced LC3 protein aggregation in the both cell types treated with C2 ceramide, in comparison with negative controls (PBS) (Figure 1). The C2 ceramide treated CD44+/CD24−/low MCF7 (P < 0.0001) and Hep-2 (P < 0.0001) cells also showed a significant increase in autophagosome areas compared to negative controls (Figure 1). These results confirmed that treatment with C2 ceramide induced autophagy in the CD44+/CD24−/low phenotype cells.
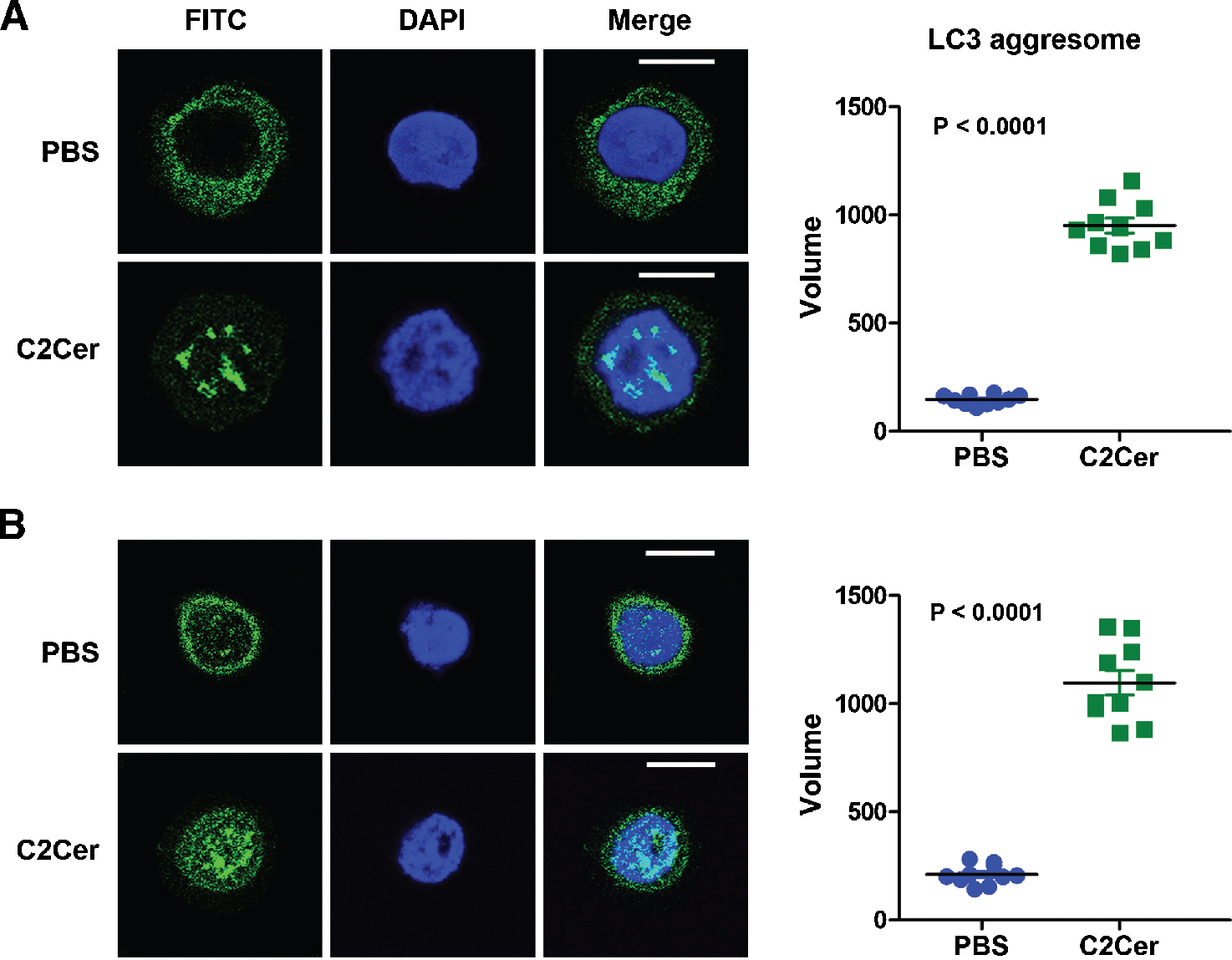 |
Figure 1. C2 ceramide induces LC3 aggregation in CD44+/CD24−/low cells. Confocal microscope image of LC3 protein stained with FITC dye by immunofluorescence assay and volume of LC3 aggresome dots in FACS sorted and then treated with 20 μM C2 ceramide (C2Cer) or PBS (as negative control) CD44+/CD24−/low phenotype (A) MCF7 and (B) Hep-2 cells indicated higher level autophagy in C2Cer treated cells in compare with negative controls. The scale of bar is 10 μm. |
To determine effect of autophagy on the CD44+/CD24−/low cell subpopulation we cultured the cells in presence of C2 ceramide for 24 hours and then analysed subpopulation of CD44+/CD24−/low phenotype in total MCF7 and Hep-2 cells. The results are shown in Figure 2. According to the results, the mean percentage of CD44+/CD24−/low subpopulation in PBS and C2 ceramide treated MCF7 cell culture were respectively 2.6 and 0.3 (P < 0.01) and in PBS and C2 ceramide treated Hep-2 cell culture were respectively 4.9 and 0.63 (P < 0.001) (Figure 2). This consequence revealed a significant reduction of CD44+/CD24−/low cell subpopulation due to autophagy in comparison with negative control (P < 0.001).
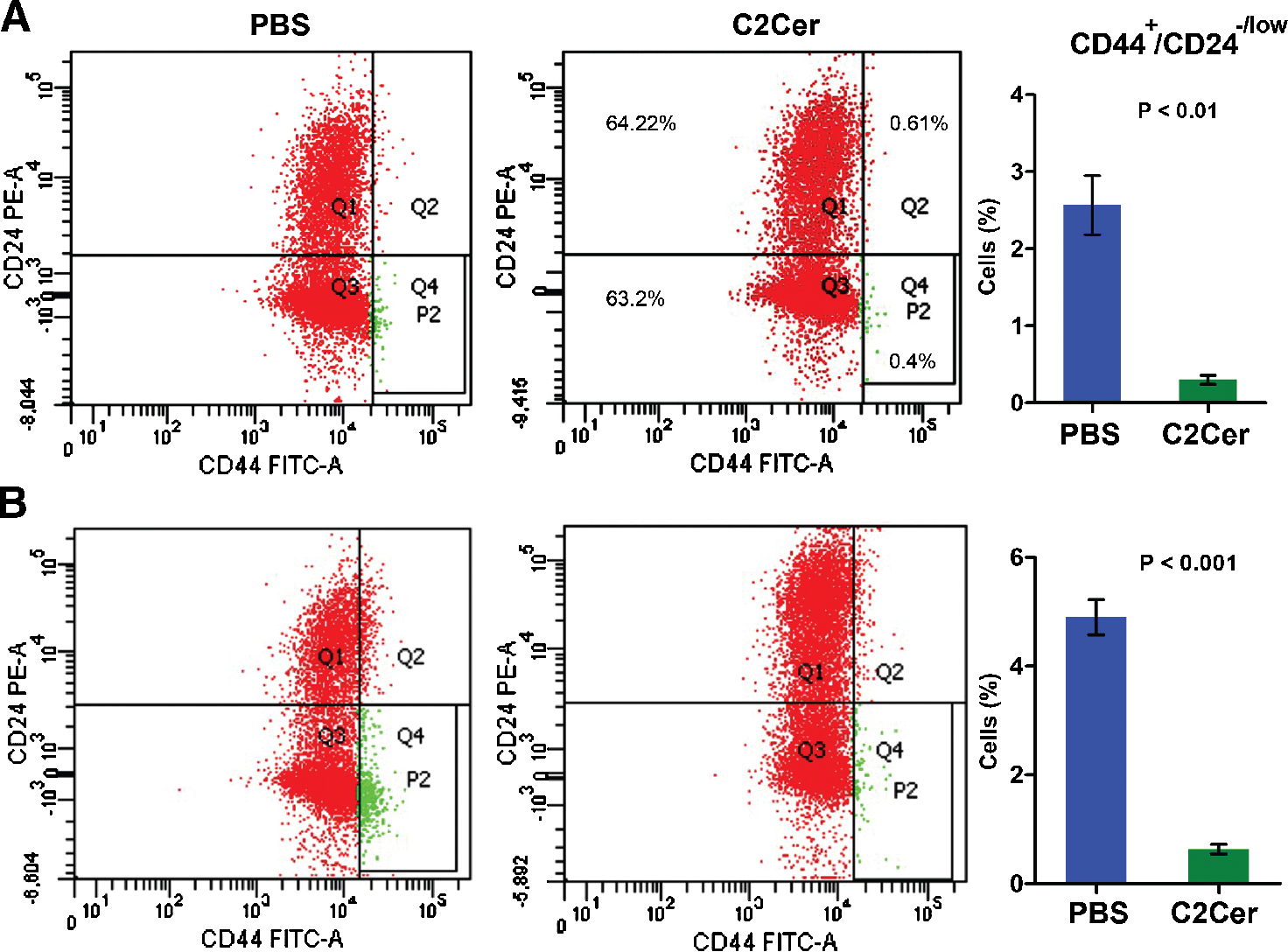 |
Figure 2. Autophagy decreases CD44+/CD24−/low cell subpopulation. Flow cytometry analysis plate and percentage of CD44+/CD24−/low phenotype (A) MCF7 and (B) Hep-2 cells considering CD44 and CD22 cell marker expression in the original cell culture were cultured in presence of 20 μM of C2 ceramide (C2Cer) or PBS (as negative control). The result indicates a significant decrease in CD44+/CD24−/low cell subpopulation under treatment with C2 cermide (P < 0.01). Quadrant 4 (panel Q4) indicates CD44+/CD24−/low cells. |
Autophagy is a catabolic regulator acts as an adaptive mechanism when intracellular and extracellular environment are poor and when cells are metabolically stressed that can ascertain the stem cells fate by impact on differentiation, survival and death [1]. Since a functionally anomaly seen in tumor cells, it appears that CSCs exploit autophagy to tumorigenesis and chemoresistance [22]. On the other way, autophagy elucidates anti-cancer properties in vitro and in vivo treatments [23] thereby, it plays two conflicting performance in tumor cells. In the present study, results showed that LC3 aggregation occurred in CD44+/CD24−/low MCF7 and Hep-2 cells due to C2 ceramide. Promotion of autophagy by C2 ceramide led to a clear increase in the number of LC3-labeled autophagosomes. LC3 is involved during the late steps of autophagy after the isolation membrane and autophagosomes has formed [24]. Here, we observed autophagy in CD44+/CD24−/low phenotype cells by monitoring LC3 aggregation. This method allows to the quantification of autophagic vesicles in each separated cell [24]. Therefore, we showed here that CSCs with CD44+/CD24−/low phenotype are efficiency susceptible to autophagy inducers. With taking into consideration a number of drugs in the clinical trials against cancers affect or act through autophagy, autophagy stimulatory approach can be considered more for discover novel anti-cancer drugs.
There has been revealed that subpopulation of CSCs in a primary tumors directly connects to resistance against chemotherapies [19], metastasis as well as malignant relapse [20]. In other words, CSC subpopulation is a positive factor for tumor regrowth, malignancy and invasion and also owing to having high DNA repair capacity and stem cell properties. The expressional evaluation of CD markers is a standard approach to identify different cell types and the assessment of their subpopulations. Hence, we used cytometry approach to obtain the scalar size of the cell subpopulation in terms of CD44 expression positive and CD24 expression negative cells. Here, evaluation of CD44 and CD24 proteins by other expression analysis likes qPCR or western blotting would not suitable method. Essentially, decrease in a stem cell subpopulation could be due to differentiation to downstream phenotypes, reduced self-renewal potency or death; however, this reduction could not be due to cell death. The challenging point here is that CD44+/CD24−/low and other phenotypic cells were equally affected by the autophagy stimulator. With regard to identical response of total cells to autophagy, it is expected equal decrease in all subpopulations and not just in CD44+/CD24−/low cells. It is currently unclear whether CSCs arise by conversion of normal stem cells or tumor cell reverse differentiation. Given that many studies demonstrated that autophagy plays a prominent role in cellular differentiation during development [1, 2], therefore changes in self-renewal and differentiation potency of the cells might be due to autophagic condition. Salemi et al. [25] reported that autophagy is an essential factor in stem cell self-renewal and differentiation; however, functional properties of CSCs are very diverse to normal stem cells. Therefore, reduction in the CSC subpopulation might be due to the possible effect of autophagy on the CSC self-renewal and alteration of their differentiation properties, although, we did not investigate here the self-renewal and differentiation of CSCs.
Recent studies revealed the positive regulatory role of autophagy in CSCs leading to survival and invasion [22, 26]. It is predicted that autophagy would be crucial for the quality control mechanisms and the maintenance of cellular homeostasis in various stem cells given their relatively long life in the organisms [27]. Additionally, very recent publications revealed effective role of autophagy induced cell death in eradiation of breast CSCs [23, 28]. Relying on our findings we suggest that MCF7 and Hep-2 derived cancer stem cells with CD44+/CD24−/low phenotype are as susceptible as the original cells against stimulation of autophagy, and the cancer stem cell subpopulation decreases due to the activation of autophagy, although the exact reason still remains unclear. We also suggest that autophagy may be a strong aim to develop CSC targeting approach that eventually allows us to discover novel CSC targeted drugs for the treatment of cancers.
The authors declare that they have no conflict of interest.
We thank Dr. Hasan Acar for providing laboratory facilities. We also thank Dr. Zeynep B Sari and Dr. Emine Yavuz from Selcuk University Advanced Technology Research and Application Center (ILTEK) for their kind aids.
| [1] | Boya P, Reggiori F, Codogno P. Emerging regulation and functions of autophagy. Nat Cell Biol 2013, 15:713–720. |
| [2] | Ryter SW, Cloonan SM, Choi AM. Autophagy: a critical regulator of cellular metabolism and homeostasis. Mol Cells 2013, 36:7–16. |
| [3] | Rao S, Tortola L, Perlot T, Wirnsberger G, Novatchkova M, Nitsch R, et al. A dual role for autophagy in a murine model of lung cancer. Nat Commun 2014, 5:3056. |
| [4] | Rosenfeldt MT, Ryan KM. The role of autophagy in tumour development and cancer therapy. Expert Rev Mol Med 2009, 11:e36. |
| [5] | Shen HM, Codogno P. Autophagic cell death: Loch Ness monster or endangered species? Autophagy 2011, 7:457–465. |
| [6] | Tsujimoto Y, Shimizu S. Another way to die: autophagic programmed cell death. Cell Death Differ 2005, 12 Suppl 2:1528–1534. |
| [7] | Degenhardt K, Mathew R, Beaudoin B, Bray K, Anderson D, Chen G, et al. Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell 2006, 10:51–64. |
| [8] | Buchser WJ, Laskow TC, Pavlik PJ, Lin HM, Lotze MT. Cell-mediated autophagy promotes cancer cell survival. Cancer Res 2012, 72:2970–2979. |
| [9] | Sui X, Chen R, Wang Z, Huang Z, Kong N, Zhang M, et al. Autophagy and chemotherapy resistance: a promising therapeutic target for cancer treatment. Cell Death Dis 2013, 4:e838. |
| [10] | Chen S, Rehman SK, Zhang W, Wen A, Yao L, Zhang J. Autophagy is a therapeutic target in anticancer drug resistance. Biochim Biophys Acta 2010, 1806:220–229. |
| [11] | Janku F, McConkey DJ, Hong DS, Kurzrock R. Autophagy as a target for anticancer therapy. Nat Rev Clin Oncol 2011, 8:528–839. |
| [12] | Wicha MS. Targeting self-renewal, an Achilles' heel of cancer stem cells. Nat Med 2014, 20:14–15. |
| [13] | Lobo NA, Shimono Y, Qian D, Clarke MF. The biology of cancer stem cells. Annu Rev Cell Dev Biol 2007, 23:675–99. |
| [14] | Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A 2003, 100:3983–3988. |
| [15] | Sun H, Jia J, Wang X, Ma B, Di L, Song G, et al. CD44+/CD24- breast cancer cells isolated from MCF-7 cultures exhibit enhanced angiogenic properties. Clin Transl Oncol 2013, 15:46–54. |
| [16] | Yan W, Chen Y, Yao Y, Zhang H, Wang T. Increased invasion and tumorigenicity capacity of CD44+/CD24- breast cancer MCF7 cells in vitro and in nude mice. Cancer Cell Int 2013, 13:62. |
| [17] | Peach RJ, Hollenbaugh D, Stamenkovic I, Aruffo A. Identification of hyaluronic acid binding sites in the extracellular domain of CD44. J Cell Biol 1993, 122:257–264. |
| [18] | Schabath H, Runz S, Joumaa S, Altevogt P. CD24 affects CXCR4 function in pre-B lymphocytes and breast carcinoma cells. J Cell Sci 2006, 119:314–325. |
| [19] | Liu H, Zhang HW, Sun XF, Guo XH, He YN, Cui SD, et al. Tamoxifen-resistant breast cancer cells possess cancer stem-like cell properties. Chin Med J (Engl) 2013, 126:3030–3034. |
| [20] | Lin Y, Zhong Y, Guan H, Zhang X, Sun Q. CD44+/CD24- phenotype contributes to malignant relapse following surgical resection and chemotherapy in patients with invasive ductal carcinoma. J Exp Clin Cancer Res 2012, 31:59. |
| [21] | Abdullah LN, Chow EK. Mechanisms of chemoresistance in cancer stem cells. Clin Transl Med 2013, 2:3. |
| [22] | Chaterjee M, van Golen KL. Breast cancer stem cells survive periods of farnesyl-transferase inhibitor-induced dormancy by undergoing autophagy. Bone Marrow Res 2011, 2011:362938. |
| [23] | Kumar D, Shankar S, Srivastava RK. Rottlerin-induced autophagy leads to the apoptosis in breast cancer stem cells: molecular mechanisms. Mol Cancer 2013, 12:171. |
| [24] | Kimura S, Fujita N, Noda T, Yoshimori T. Monitoring autophagy in mammalian cultured cells through the dynamics of LC3. Methods Enzymol 2009, 452:1–12. |
| [25] | Salemi S, Yousefi S, Constantinescu MA, Fey MF, Simon HU. Autophagy is required for self-renewal and differentiation of adult human stem cells. Cell Res 2012, 22:432–435. |
| [26] | Gong C, Bauvy C, Tonelli G, Yue W, Deloménie C, et al. Beclin 1 and autophagy are required for the tumorigenicity of breast cancer stem-like/progenitor cells. Oncogene 2013, 32:2261–2272. |
| [27] | Guan JL, Simon AK, Prescott M, Menendez JA, Liu F, Wang F, et al. Autophagy in stem cells. Autophagy 2013, 9:830–849. |
| [28] | Jain K, Paranandi KS, Sridharan S, Basu A. Autophagy in breast cancer and its implications for therapy. Am J Cancer Res 2013, 3:251–265. |