| Journal of Cancer Stem Cell Research (2014), 2:e1002 © 2013 Creative Commons. All rights reserved ISSN 2329-5872 DOI: 10.14343/JCSCR.2014.2e1002 http://cancerstemcellsresearch.com |
 |
| Journal of Cancer Stem Cell Research (2014), 2:e1002 © 2013 Creative Commons. All rights reserved ISSN 2329-5872 DOI: 10.14343/JCSCR.2014.2e1002 http://cancerstemcellsresearch.com |
 |
| Review Article | Open Access |
| Understanding the biological functions and therapeutic potentials of stem cells and cancer stem cells: Where are we? | |
| Dhruv Kumar1, Sharmila Shankar*2,3, Rakesh K. Srivastava*1 | |
| 1Department of Pharmacology, Toxicology and Therapeutics, and Medicine, The University of Kansas Medical Center, 3901 Rainbow Boulevard, Kansas City, KS, 66160, USA, 2Kansas City VA Medical Center, 4801 Linwood Boulevard, Kansas City, MO 64128, 3Department of Pathology, School of Medicine, University of Missouri, Kansas City, Missouri. | |
| *Correspondence: Rakesh K. Srivastava, Department of Pharmacology, Toxicology and Therapeutics, and Medicine, The University of Kansas Medical Center, 3901 Rainbow Boulevard, Kansas City, KS, 66160, USA. Phone: 913-945-6686; E-mail: rsrivastava@kumc.edu Received: April 20, 2014; Revised: May 1, 2014; Accepted: May 2, 2014 | |
Abstract: In the recent years, cancer stem cell research has evolved as one of the most significant ways to resolve the cancer therapeutics problem. In this review, we will discuss the importance of cancer stem cells in current therapeutic approaches. The idea of cancer stem cells was originally evolved from the normal stem cells which are known as building blocks of life. It has been clear for long time that some cells had the ability to produce other type of cells. In recent years cancer stem cell research has progressed dramatically and large number of research studies are published each year in scientific journals. Progress in the cancer stem cell research have made possible to develop novel strategies to treat several malignancies and become hot topic of discussion. Development of potential personalized medicine and its clinical applications for treatment of human cancers are ultimate goal of most cancer stem cell based therapy.
Keywords: Cancer Stem Cells, Stem Cells, Translational Research, Cancer
In recent years, stem cell research has evolved as one of the fastest growing research area in the field of development of therapeutics. Stem cells are undifferentiated/unspecialized cells that have the ability for self-renewal and generate highly specific cells endowed with specific function. Human body contains more than 200 different types of cells that are organized into tissues and organs with highly specific functions. The origins of stem cell research bring us closer to understand how tissues are maintained in adult life. Long time ago it was proposed that the differentiated cells which forms structurally and functionally different tissues, such as the skin, blood and intestinal epithelium, have a short lifespan and are unable to renew themselves. This directed to the concept that such tissues are maintained by cells with extensive renewal capacity and the ability to regenerate that can undergo further differentiation called “stem cells” [1]. Such cells generate only the differentiated families which are appropriate for the tissue in which they exist and are thus referred to as multipotent/unipotent stem cells (Fig. 1). Initially, stem cell research was focused on only three types of tissue: such as epidermis, with rapid turnover of differentiated cells; brain, in which there appear to be no self-renewal; and liver, in which cells divide to give two daughter cells that were functionally equivalent [2]. While it remains true that different adult tissues differ in terms of the proportion of proliferative cells and the nature of the differentiation compartment. In recent years it has become apparent that some tissues that appeared to lack self-renewal ability do indeed contain stem cells [3] and others contain a previously unrecognized cellular heterogeneity [4]. These cells have received widespread attention in the recent years [5–7]. The stem cell concept is important because it promises new therapies against several diseases (Fig. 2).
 |
Figure 1. Generation of different tissues from stem cells. Pluripotent stem cells can form many different types of adult organism. Totipotent stem cells can form extra embryonic tissues of the embryo. |
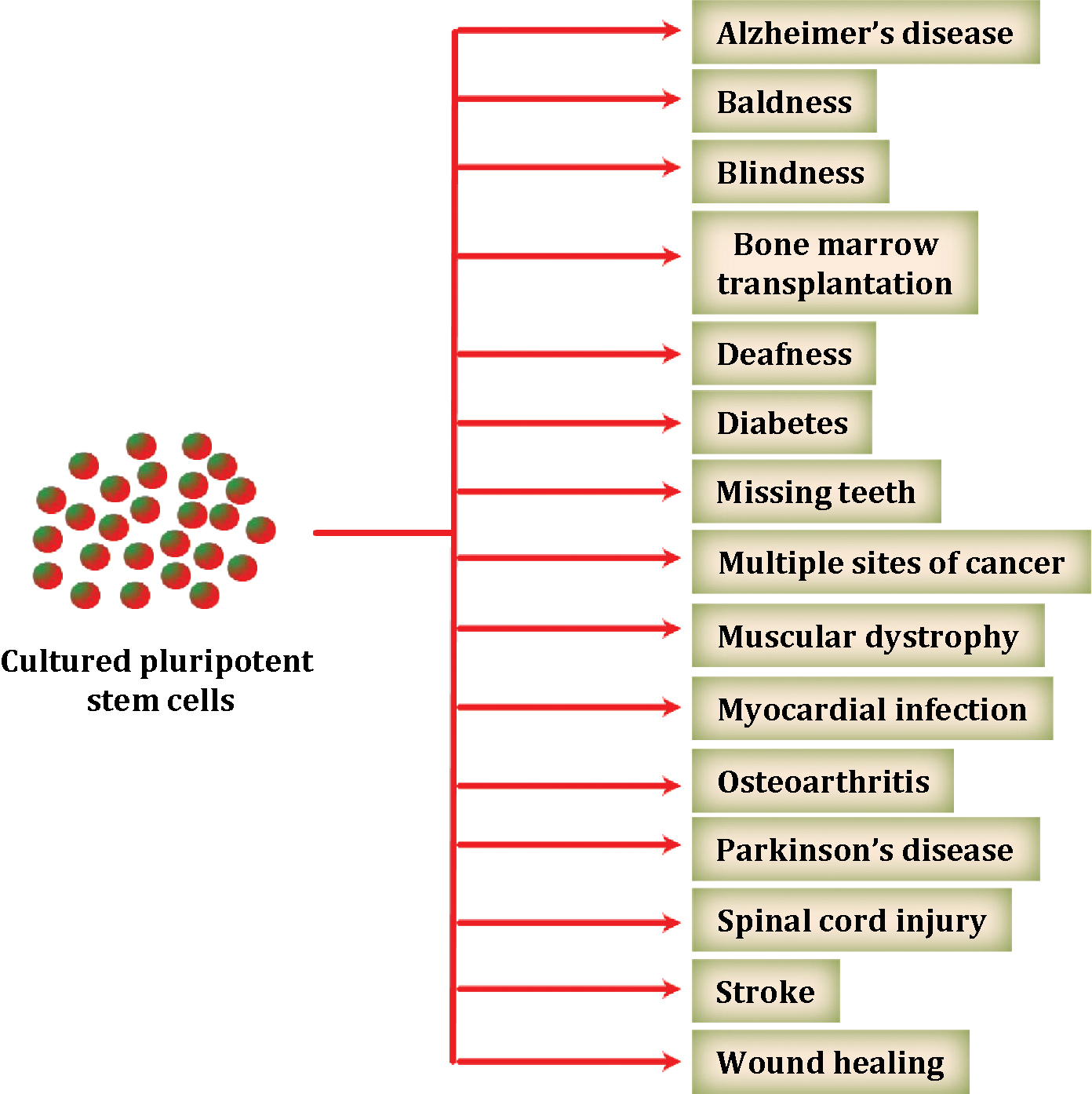 |
Figure 2. Stem cells can be used to treat several diseases. Stem cells are undifferentiated cells which when placed into appropriate niches or when driven by external factors (e.g. growth factors, cytokines, matrix) may differentiate into distinct organs. Potentially, stem cells can be used to repair damages in organs and tissue, treat neurodegenerative disease, heart disease, several cancers, diabetes, etc. |
Stem cells are define as “the undifferentiated cells which are found in the body that have potential to self-renew and develop into many different cell types during early life and growth which are structurally and functionally different from each other”. Until now, scientists mainly worked with two types of stem cells from animals and humans: embryonic stem cells and non-embryonic somatic or adult stem cells. In 1998 stem cells were derived from human embryo and cultured in laboratory, these cells are called human embryonic stem cells. In 2006, researchers were successfully “reprogrammed” some specialized adult cells that genetically “reprogrammed” cells and were assumed a stem cell-like state. This new type of stem cell, called induced pluripotent stem cells.
Stem cells research is rapidly expanding. Stem cells have remarkable potential to develop into many different cell types in the human body. It has been found that stem cells serve as a sort of internal repair system, dividing continously to replace cells that undergo senescence as long as the person or animal is alive. Stem cells divide to form either a new stem cell or become other cell types with a more specialized function. Stem cells can be distinguished from other cell types such as unspecialized cells capable of renewing themselves through cell division, sometimes after long periods of inactivity, and under certain physiologic or experimental conditions, they can be induced to become tissue- or organ-specific cells with special functions.
Stem cells are important for living organisms for many reasons. In the 3- to 5-day-old murine embryo, called a blastocyst, the inner cells give rise to the entire body of the organism, including all specialized cell types and organs such as the lung, heart, skin, eggs, sperm and other tissues. In some adult tissues, such as bone marrow, muscle, and brain, discrete populations of adult stem cells are responsible for regeneration or replacements for cells that are lost through normal wear and tear, injury, or disease. Given their unique regenerative abilities, stem cells provide a new potential for treating several diseases such as cancer, diabetes, stroke, and heart disease. Research work is in progress to screen new drugs and to develop novel model systems by applying stem cells to study normal growth and to identify the cause of birth defects.
The first evidence for the role of stem cells in cancer was provided by Dick in 1994 [8] in human acute myeloid leukemia (AML), in which AML-initiating cell population was identified in patients by transplantation of these cells into severe combined immune-deficient (SCID) mice. The leukemia-initiating cells were enriched on the basis of cell surface marker expression (CD34+/CD38−). In 2003, human CSCs were identified in solid tumors, including breast [8] and brain cancer [9]. The successive reports identified CSCs in a variety of tumors, including colon, lung, prostate, pancreas, glioblastoma, and melanoma (
| Table 1. Phenotype of cancer stem cell/cancer cells in various human solid malignancies. | ||||
| S.No. | Cancer type | Phenotype | Xenograft model used | References |
|---|---|---|---|---|
| 1 | Brain | CD133+ | NOD/SCID | [46–48] |
| CD133+ | nu/nu | [49, 50] | ||
| SSEA-1, SOX2 | NOD/SCID | [51] | ||
| Nestin | pNestin-GFP transgenic mice | [52] | ||
| CD44+CD9+CD66+ | Glioma Mouse Model | [53] | ||
| 2 | Breast | CD44+CD24− Lin− | NOD/SCID | [8, 54–56] |
| ALDH1+ | NOD/SCID | [10, 56] | ||
| 3 | Colon | CD133+ | NOD/SCID | [57] |
| CD44+/Ep-CAM+ | NOD/SCID | [58, 59] | ||
| CD110+CDCP1+ | Thrombopoietin null mice (TPO−/−) | [60] | ||
| 4 | Head and neck | CD44+ Cytokeratin 5/14+ | NOD/SCID | [61] |
| 5 | Liver | CD90+CD44+ | SCID/Beige, BALB/c | [62] |
| 6 | Lung | CD133+Ep-CAM+ | NOD/SCID | [63] |
| 7 | Thyroid | NANOG, OCT4 | NOD/SCIDII2rg−/− | [50] |
| CD133+ | NOD/SCID | [64] | ||
| 8 | Melanoma | ABCB5+ | NOD/SCID | [65] |
| 1:4 unselected cells | NOD/SCID/IL2Rγ- | [66] | ||
| 9 | Pancreas | ALDH1+ | NOD/SCID | [67] |
| CD133+ | NMRI-nu/nu | [12, 68] | ||
| CD44+CD24+ESA+ | NOD/SCID | [12, 69] | ||
| DCLK1 | Wild-Type (WT) C57BL/6 mice | [70, 71] | ||
| 10 | Prostate | CD133+ | NOD/SCID | [72] |
| CD44+ | NOD/SCID | [73] | ||
| CD44+/α2β1+/CD133+ | Methylcellulose progenitor assay | [74] | ||
An alternative theory for the origin of CSCs suggests that they arise from normal somatic cells which acquire stem-like characteristics and malignant behavior through genetic and/or heterotypic alterations. For example, cancer cells gain stem-like characteristics through epithelial-mesenchymal transition (EMT). The induction of EMT in immortalized human mammary epithelial cells (HMLEs) resulted in the acquisition of mesenchymal traits and expression of stem-cell markers, which are similar to those stem cell-like cells isolated from HMLE. Furthermore, these cells have an increased ability to form mammospheres [14]. EMT is driven by transcription factors, including SNAIL1/2, SLUG, ZEB1/2, or TWIST1/2, which increase the invasiveness of epithelial cells. In several studies, the induction of EMT enhances self-renewal and the acquisition of CSC characteristics [14–16].
Stem cells have unique identification markers that allow thir purification from other cells in the body. All stem cells are capable of dividing and renewing themselves for long periods, are unspecialized and can give rise to specialized cell types. It has taken many years to learn how to derive and maintain stem cells in the laboratory without spontaneously differentiating them into specific cell types. Stem cells are unspecialized. One of the unique properties of a stem cell is that it does not have any tissue-specific structures that allow it to perform specialized functions. For example, a stem cell cannot work with its neighbors to pump blood through the body (like a heart muscle cell), and it cannot carry oxygen molecules through the bloodstream (like a red blood cell). However, unspecialized stem cells can give rise to specialized cells, including heart muscle cells, blood cells, or nerve cells.
Adult stem cells typically generate the cell types of the tissue in which they reside. For example, a blood-forming adult stem cell in the bone marrow normally gives rise to the many types of blood cells. It is generally accepted that a blood-forming cell in the bone marrow which is called a hematopoietic stem cells cannot give rise to the cells of a very different tissue, such as nerve cells in the brain. It has been shown that stem cells from one tissue/organ may give rise to cell type of a completely different tissue/organ (Fig. 1). However, it remains an area of great debate within the research community.
It is important to follow the mechanisms by which stem cells are useful in treatment and how stem cells translocate to the specific sites to benefit patients in the clinics [17]. The hematopoietic stem cell transplantation is one of the earliest stem cell therapies and is most widely used [18]. Mainly the stem cells come from bone marrow, peripheral blood or cord blood. For some applications, the patients' own cells are transplanted. However, allogeneic stem cell transplantation is now a common procedure for the treatment of bone marrow failure and hematological malignancies, such as leukemia. Donor stem cells are used to reconstitute immune function in such patients following radiation and/or chemotherapy.
Clinical studies over the last 10 years suggest that stem cell transplantation also has potential as a therapy for neurodegenerative diseases. Stem cells reprograming has been shown to treat Parkinsons's disease and brain related disorders [19]. Clinical trials have been conducted for grafting of brain tissue from aborted fetuses into patients with Parkinson's disease and Huntington's disease [20, 21]. In such studies, some successes have been noted; however, outcomes have not been uniform and will require further clinical trials involving more refined patient selection, in an attempt to predict better outcome. Noticeably, aside from the opposition in many quarters to using fetal material, there are practical challenges associated with availability and regularity of the grafted cells. Therapies with pure populations of stem cells are important and achievable [22, 23]. Stem cells definitely offer great potential to treat many human diseases such as tissue repair resulting from injury or ageing.
Importance aspects of stem cells are that these cells are like blank paper; we can assign task to stem cells, and modify them to become a part of any tissue/organ. Using stem cell therapy, several successful treatments for many diseases related to liver, bladder, ear, lung, brain, heart a few to name have been reported [17, 19]. Recently, stem cell therapy has also shown potential cure against AIDS [24, 25].
Even though, stem cell based therapies are not new, but this new approach has long way to go. The first successful stem-cell based therapy was performed to treat leukemia by bone marrow transplantation [26]. In patient with damaged bone marrow as a result of radiation and/or chemotherapy, donor bone marrow cells were injected into the patient and the bone marrow stem cells established themselves in the patient's bones. The donor bone marrow cells differentiated into blood cells that the patient needed. Often, the patient needs drugs to prevent his or her immune system from rejecting the new bone marrow [27], but this procedure uses existing hematopoietic stem cells. Stem cells can also be used to treat a failing heart by stimulating stem cells into differentiated heart cells and by injecting them into the patient's damaged heart, and the new heart cells could grow and repair the damaged tissue [17]. Finally, by studying how stem cells differentiate into specialized cells, will help to understand birth defects and ways to prevent or treat such defects.
The conventional treatment of cancer is to use chemotherapy. “The ideal anti-cancer drug should be one that has a specific affinity for cancer cells without affecting normal cells” [28]. During World War I, physicians hypothesized that mustard agents may therefore be able to treat leukemia, which is a disease caused by abnormal proliferation of myeloid or lymphoid precursor cells. In next several decades we observed a rapid increase of new therapeutic agents, including anti-metabolites, DNA damaging agents, and taxanes, as well as the introduction of combination therapy. Most efforts for cancer therapy involve direct interference of cell proliferation by altering events that occur during the cell cycle, as cell growth in cancer cell is largely unregulated. Early chemotherapy focused on inhibiting cell growth through mitotic poisons to control tumor cell proliferation [29–31]. However, later research also exposed alterations in vasculature, growth regulation, and evasion of cell death as essential events in tumor growth [29, 32, 33]. Such changes present additional targets for anti-cancer strategies. We present here a brief overview of cancer treatments and discuss how translational research has led to the development of targeted therapies.
Ionizing radiation and chemotherapy both are important cancer therapies that have been widely used since their efficacy was first demonstrated over a century ago. Ionizing radiation utilizes high-energy radiation to kill cancer cells by inducing lethal DNA damage and is often used in conjunction with either surgery or chemotherapy [34, 35]. Although radiation therapy is generally well tolerated, secondary cancers, skeletal complications, radiation-induced heart disease, and lung disease are common side effects [36, 37]. Due to the toxicity of radiation, much focus has been placed on improving its cancer cell specificity. This includes research of agents that sensitize cancer cells to radiation or protect normal cells from damage induced by radiation [38–40]. Chemotherapy involves the use of systemic drugs targeting of various aspects of cell growth. Chemotherapy agents vary widely in their chemical composition, function, specificity, and toxicity. While generally effective, chemotherapeutic agents are highly toxic and damage normal cells as well as cancer cells, causing severe side effects. New lines of chemotherapies to be used in both solo and combination therapy are being developed to increase treatment efficacy and reduce side effects associated with each drug. Since, many drugs are not administered easily into patients or reach in sufficient amount to cancer, therefore, various drug delivery systems that can deliver high pay load of drug to cancer are being developed. Side effects of chemotherapy can be devastating to a patient, therefore, chemoprotective and chemosensitizing agents are being developed to target cancer stem cells to increase drug efficacy and to reduce side effects. [30, 31].
A key success for the development of a highly efficacious and effective drug is development of a molecular targeted drug, which targets cancer cell growth inhibited by interfering with specific molecules that are necessary for tumor growth. Identification of specific molecular characteristics of tumors has facilitated rational drug designing, and recognizing suitable targets has led to the generation of countless compounds that have been highly effective in both preclinical and clinical trials. These compounds are screened based on their effects on specific targets in tumor cells and the overall effect on cancer cell growth [41].
Stem cells have potential to treat many human diseases, including ageing, cancer, diabetes, blindness, heart failure and neurodegeneration [17, 19, 42]. Nevertheless, it is essential to be realistic about the time and steps required to take new therapies into the clinic. It is inspiring to be able to induce embryonic stem cells to differentiate into cardiomyocytes in a culture dish, but that is only one very small step towards effecting cardiac repair. At present, there are a large number of human embryonic stem cell lines available for clinical application offering the opportunity for optimal immunological matching of donors and recipients. However, one of the attractions of transplanting stem cells is use of patient's own cells, avoiding the need for immunosuppression. This invention will help us to understand how the pluripotent state can be efficiently and stably induced and maintained by treating cells with pharmacologically active compounds rather than by genetic manipulation [43]. Recently, it has been shown that pancreatic exocrine cells in adult mice can be reprogrammed to become functional, insulin-producing beta cells by expression of transcription factors that regulate pancreatic development [42, 44]. The idea of repairing tissue through a process of cellular reprogramming in situ is an attractive paradigm to be explored. A range of biomaterials are already in clinical use for tissue repair, in particular to repair defects in cartilage and bone [45]. Moreover, finding drugs that selectively target cancer stem cells offers the potential to develop cancer treatments that are not only more effective, but also cause less collateral damage to the patient's normal tissues and prevent recurrence of cancer. In addition, patient-specific iPS cells provide a new tool to identify underlying disease mechanisms.
The field of stem cell and cancer stem cell research has entered in inspiring translational phase aiming to produce discoveries that will be applicable for regenerative medicine to treat several diseases. Focus on stem cell research and the transition of current therapy to stem-cell-based therapy will help to develop new therapeutics that will be more specific and efficacious for the treatment of cancer as well as many other diseases with the potential to reduce side effects as well recurrence of disease. The capacity to reprogram human adult somatic cells into a pluripotent state or a different cell type will open the door to the development and study of disease-specific cells. These cells not only will provide a tool to understand more about the mechanistic basis of disease, but also offers the possibility of drug testing in the lab. Although clinical trials using reprogrammed patient-specific cells are a long way to go, but other areas of stem cell research are closer to clinical application.
We would like to thank all the lab members for critical reading of this review article. The project was funded through the National Institutes of Health (RKS), Kansas Bioscience Authority (RKS) and The VA Merit Award (SS).
| [1] | Lajtha LG. Stem cell concepts. Nouv Fr Hematol 1979, 21:59–65. |
| [2] | Hall PA, Watt FM. Stem cells: the generation and maintenance of cellular diversity. Development 1989, 106:619–33. |
| [3] | Zhao C, Deng W, Gage FH. Mechanisms and functional implications of adult neurogenesis. Cell 2008, 132:645–60. |
| [4] | Zaret KS, Grompe M. Generation and regeneration of cells of the liver and pancreas. Science 2008, 322:1490–4. |
| [5] | Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature 2001, 414:105–11. |
| [6] | Clarke MF, Dick JE, Dirks PB, Eaves CJ, Jamieson CH, Jones DL, Visvader J, Weissman IL, Wahl GM. Cancer stem cells–perspectives on current status and future directions: AACR Workshop on cancer stem cells. Cancer Res 2006, 66:9339–44. |
| [7] | Dick JE. Stem cell concepts renew cancer research. Blood 2008, 112:4793–807. |
| [8] | Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A 2003, 100:3983–8. |
| [9] | Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C, Squire J, Dirks PB. Identification of a cancer stem cell in human brain tumors. Cancer Res 2003, 63:5821–8. |
| [10] | Ginestier C, Hur MH, Charafe-Jauffret E, Monville F, Dutcher J, Brown M, Jacquemier J, Viens P, Kleer CG, Liu S, Schott A, Hayes D, Birnbaum D, Wicha MS, Dontu G. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 2007, 1:555–67. |
| [11] | Yu Z, RG P. microRNAs and Cancer Stem Cells. In: Cho WCS, editor. MicroRNAs in Cancer Translational Research. Springer, 2011, P373–88. |
| [12] | Shankar S, Nall D, Tang SN, Meeker D, Passarini J, Sharma J, Srivastava RK. Resveratrol inhibits pancreatic cancer stem cell characteristics in human and KrasG12D transgenic mice by inhibiting pluripotency maintaining factors and epithelial-mesenchymal transition. PLoS One 2011, 6:e16530. |
| [13] | Kelly PN, Dakic A, Adams JM, Nutt SL, Strasser A. Tumor growth need not be driven by rare cancer stem cells. Science 2007, 317:337. |
| [14] | Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, Campbell LL, Polyak K, Brisken C, Yang J, Weinberg RA. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008, 133:704–15. |
| [15] | Ansieau S, Bastid J, Doreau A, Morel AP, Bouchet BP, Thomas C, Fauvet F, Puisieux I, Doglioni C, Piccinin S, Maestro R, Voeltzel T, Selmi A, Valsesia-Wittmann S, Caron de Fromentel C, Puisieux A. Induction of EMT by twist proteins as a collateral effect of tumor-promoting inactivation of premature senescence. Cancer Cell 2008, 14:79–89. |
| [16] | Yang J, Mani SA, Donaher JL, Ramaswamy S, Itzykson RA, Come C, Savagner P, Gitelman I, Richardson A, Weinberg RA. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 2004, 117:927–39. |
| [17] | Maltais S, Joggerst SJ, Hatzopoulos A, Disalvo TG, Zhao D, Sung HJ, Wang X, Byrne JG, Naftilan AJ. Stem cell therapy for chronic heart failure: an updated appraisal. Expert Opin Biol Ther 2013. |
| [18] | Austin EB, Guttridge M, Pamphilon D, Watt SM. The role of blood services and regulatory bodies in stem cell transplantation. Vox Sanguinis 2008, 94:6–17. |
| [19] | Ali F, Stott SR, Barker RA. Stem cells and the treatment of Parkinson's disease. Exp Neurol 2013. |
| [20] | Dunnett SB, Bjorklund A, Lindvall O. Cell therapy in Parkinson's disease - stop or go? Nature Reviews Neuroscience 2001, 2:365–69. |
| [21] | Wright BLC, Barker RA. Established and emerging therapies for Huntington's disease. Current Molecular Medicine 2007, 7:579–87. |
| [22] | Conti L, Pollard SM, Gorba T, Reitano E, Toselli M, Biella G, Sun YR, Sanzone S, Ying QL, Cattaneo E, Smith A. Niche-independent symmetrical self-renewal of a mammalian tissue stem cell. Plos Biology 2005, 3:1594–606. |
| [23] | Lowell S, Benchoua A, Heavey B, Smith AG. Notch promotes neural lineage entry by pluripotent embryonic stem cells. Plos Biology 2006, 4:805–18. |
| [24] | Hutter G, Nowak D, Mossner M, Ganepola S, Mussig A, Allers K, Schneider T, Hofmann J, Kucherer C, Blau O, Blau IW, Hofmann WK, Thiel E. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. N Engl J Med 2009, 360:692–8. |
| [25] | Allers K, Hutter G, Hofmann J, Loddenkemper C, Rieger K, Thiel E, Schneider T. Evidence for the cure of HIV infection by CCR5Delta32/Delta32 stem cell transplantation. Blood 2011, 117:2791–9. |
| [26] | Gluckman E, Broxmeyer HA, Auerbach AD, Friedman HS, Douglas GW, Devergie A, Esperou H, Thierry D, Socie G, Lehn P, |
| [27] | Rubinstein P, Carrier C, Scaradavou A, Kurtzberg J, Adamson J, Migliaccio AR, Berkowitz RL, Cabbad M, Dobrila NL, Taylor PE, Rosenfield RE, Stevens CE. Outcomes among 562 recipients of placental-blood transplants from unrelated donors. New England Journal of Medicine 1998, 339:1565–77. |
| [28] | Beumer HM, Porton WM. Morphologic effects of cytotoxic drugs on tumor cells. Dis Chest 1968, 53:252–6. |
| [29] | Leighton J. Propagation of cancer: targets for future chemotherapy. Cancer Res 1969, 29:2457–65. |
| [30] | Scott RB. Cancer chemotherapy–the first twenty-five years. Br Med J 1970, 4:259–65. |
| [31] | DeVita VT Jr. Chu E. A history of cancer chemotherapy. Cancer Res 2008, 68:8643–53. |
| [32] | Chabner BA, Boral AL, Multani P. Translational research: walking the bridge between idea and cure–seventeenth Bruce F. Cain Memorial Award lecture. Cancer Res 1998, 58:4211–6. |
| [33] | Hanahan D, Weinberg RA. The hallmarks of cancer. Cell 2000, 100:57–70. |
| [34] | Bernier J, Hall EJ, Giaccia A. Radiation oncology: a century of achievements. Nat Rev Cancer 2004, 4:737–47. |
| [35] | Haffty BG, Kim JH, Yang Q, Higgins SA. Concurrent chemo-radiation in the conservative management of breast cancer. Int J Radiat Oncol Biol Phys 2006, 66:1306–12. |
| [36] | Hirbe A, Morgan EA, Uluckan O, Weilbaecher K. Skeletal complications of breast cancer therapies. Clin Cancer Res 2006, 12:6309s–6314s. |
| [37] | Munshi A. Breast cancer radiotherapy and cardiac risk: the 15-year paradox. J Cancer Res Ther 2007, 3:190–2. |
| [38] | Torres-Roca JF, Stevens CW. Predicting response to clinical radiotherapy: past, present, and future directions. Cancer Control 2008, 15:151–6. |
| [39] | Sofou S. Radionuclide carriers for targeting of cancer. Int J Nanomedicine 2008, 3:181–99. |
| [40] | van Meerbeeck JP, Meersschout S, De Pauw R, Madani I, De Neve W. Modern radiotherapy as part of combined modality treatment in locally advanced non-small cell lung cancer: present status and future prospects. Oncologist 2008, 13:700–8. |
| [41] | Saijo N, Nishio K, Tamura T. Translational and clinical studies of target-based cancer therapy. Int J Clin Oncol 2003, 8:187–92. |
| [42] | Nostro MC, Keller G. Generation of beta cells from human pluripotent stem cells: Potential for regenerative medicine. Semin Cell Dev Biol 2012, 23:701–10. |
| [43] | Silva J, Barrandon O, Nichols J, Kawaguchi J, Theunissen TW, Smith A. Promotion of reprogramming to ground state pluripotency by signal inhibition. Plos Biology 2008, 6:e253. |
| [44] | Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature 2008, 455:627–32. |
| [45] | Kamitakahara M, Ohtsuki C, Miyazaki T. Review paper: behavior of ceramic biomaterials derived from tricalcium phosphate in physiological condition. J Biomater Appl 2008, 23:197–212. |
| [46] | Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, Henkelman RM, Cusimano MD, Dirks PB. Identification of human brain tumour initiating cells. Nature 2004, 432:396–401. |
| [47] | Lenkiewicz M, Li N, Singh SK. Culture and isolation of brain tumor initiating cells. Curr Protoc Stem Cell Biol 2009, Chapter 3: Unit3.3. |
| [48] | Liu Q, Nguyen DH, Dong Q, Shitaku P, Chung K, Liu OY, Tso JL, Liu JY, Konkankit V, Cloughesy TF, Mischel PS, Lane TF, Liau LM, Nelson SF, Tso CL. Molecular properties of CD133+ glioblastoma stem cells derived from treatment-refractory recurrent brain tumors. J Neurooncol 2009, 94:1–19. |
| [49] | Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, Dewhirst MW, Bigner DD, Rich JN. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006, 444:756–60. |
| [50] | Pavon LF, Marti LC, Sibov TT, Miyaki LA, Malheiros SM, Mamani JB, Brandt RA, Ribas GC, Pagura JR, Joaquim MA, Feres Junior H, Gamarra LF. Isolation, cultivation and characterization of CD133+ stem cells from human glioblastoma. Einstein (Sao Paulo) , 2012, 10:197–202. |
| [51] | Son MJ, Woolard K, Nam DH, Lee J, Fine HA. SSEA-1 Is an Enrichment Marker for Tumor-Initiating Cells in Human Glioblastoma. Cell Stem Cell 2009, 4:440–52. |
| [52] | Matsumura S, Takagi K, Okuda-Ashitaka E, Lu J, Naritsuka H, Yamaguchi M, Ito S. Characterization of nestin expression in the spinal cord of GFP transgenic mice after peripheral nerve injury. Neuroscience 2010, 170:942–53. |
| [53] | Behnan J, Isakson P, Joel M, Cilio C, Langmoen IA, Vik-Mo EO, Badn W. Recruited brain tumor-derived mesenchymal stem cells contribute to brain tumor progression. Stem Cells 2014, 32:1110–23. |
| [54] | Marsden CG, Wright MJ, Pochampally R, Rowan BG. Breast tumor-initiating cells isolated from patient core biopsies for study of hormone action. Methods Mol Biol 2009, 590:363–75. |
| [55] | Dontu G, Liu SL, Wicha MS. Stem cells in mammary development and carcinogenesis - Implications for prevention and treatment. Stem Cell Reviews 2005, 1:207–13. |
| [56] | Ricardo S, Vieira AF, Gerhard R, Leitao D, Pinto R, Cameselle-Teijeiro JF, Milanezi F, Schmitt F, Paredes J. Breast cancer stem cell markers CD44, CD24 and ALDH1: expression distribution within intrinsic molecular subtype. Journal of Clinical Pathology 2011, 64:937–46. |
| [57] | O'Brien CA, Pollett A, Gallinger S, Dick JE. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 2007, 445:106–10. |
| [58] | Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C, De Maria R. Identification and expansion of human colon-cancer-initiating cells. Nature 2007, 445:111–5. |
| [59] | Kimura Y, Goi T, Nakazawa T, Hirono Y, Katayama K, Urano T, Yamaguchi A. CD44variant exon 9 plays an important role in colon cancer initiating cells. Oncotarget 2013, 4:785–91. |
| [60] | Gao W, Chen L, Ma Z, Du Z, Zhao Z, Hu Z, Li Q. Isolation and phenotypic characterization of colorectal cancer stem cells with organ-specific metastatic potential. Gastroenterology 2013, 145:636–46 e5. |
| [61] | Prince ME, Sivanandan R, Kaczorowski A, Wolf GT, Kaplan MJ, Dalerba P, Weissman IL, Clarke MF, Ailles LE. Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc Natl Acad Sci U S A 2007, 104:973–8. |
| [62] | Yang ZF, Ho DW, Ng MN, Lau CK, Yu WC, Ngai P, Chu PW, Lam CT, Poon RT, Fan ST. Significance of CD90+ cancer stem cells in human liver cancer. Cancer Cell 2008, 13:153–66. |
| [63] | Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni M, Di Virgilio A, Conticello C, Ruco L, Peschle C, De Maria R. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ 2008, 15:504–14. |
| [64] | Friedman S, Lu M, Schultz A, Thomas D, Lin RY. CD133+ anaplastic thyroid cancer cells initiate tumors in immunodeficient mice and are regulated by thyrotropin. PLoS One 2009, 4:e5395. |
| [65] | Schatton T, Murphy GF, Frank NY, Yamaura K, Waaga-Gasser AM, Gasser M, Zhan Q, Jordan S, Duncan LM, Weishaupt C, Fuhlbrigge RC, Kupper TS, Sayegh MH, Frank MH. Identification of cells initiating human melanomas. Nature 2008, 451:345–9. |
| [66] | Quintana E, Shackleton M, Sabel MS, Fullen DR, Johnson TM, Morrison SJ. Efficient tumour formation by single human melanoma cells. Nature 2008, 456:593–8. |
| [67] | Rasheed ZA, Yang J, Wang Q, Kowalski J, Freed I, Murter C, Hong SM, Koorstra JB, Rajeshkumar NV, He X, Goggins M, Iacobuzio-Donahue C, Berman DM, Laheru D, Jimeno A, Hidalgo M, Maitra A, Matsui W. Prognostic significance of tumorigenic cells with mesenchymal features in pancreatic adenocarcinoma. J Natl Cancer Inst 2010, 102:340–51. |
| [68] | Hermann PC, Huber SL, Herrler T, Aicher A, Ellwart JW, Guba M, Bruns CJ, Heeschen C. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell 2007, 1:313–23. |
| [69] | Li C, Heidt DG, Dalerba P, Burant CF, Zhang L, Adsay V, Wicha M, Clarke MF, Simeone DM. Identification of pancreatic cancer stem cells. Cancer Res 2007, 67:1030–7. |
| [70] | Bailey JM, Alsina J, Rasheed ZA, McAllister FM, Fu YY, Plentz R, Zhang H, Pasricha PJ, Bardeesy N, Matsui W, Maitra A, Leach SD. DCLK1 marks a morphologically distinct subpopulation of cells with stem cell properties in preinvasive pancreatic cancer. Gastroenterology 2014, 146:245–56. |
| [71] | Saqui-Salces M, Keeley TM, Grosse AS, Qiao XT, El-Zaatari M, Gumucio DL, Samuelson LC, Merchant JL. Gastric tuft cells express DCLK1 and are expanded in hyperplasia. Histochem Cell Biol 2011, 136:191–204. |
| [72] | Patrawala L, Calhoun T, Schneider-Broussard R, Zhou J, Claypool K, Tang DG. Side population is enriched in tumorigenic, stem-like cancer cells, whereas ABCG2+ and ABCG2- cancer cells are similarly tumorigenic. Cancer Res 2005, 65:6207–19. |
| [73] | Patrawala L, Calhoun T, Schneider-Broussard R, Li H, Bhatia B, Tang S, Reilly JG, Chandra D, Zhou J, Claypool K, Coghlan L, Tang DG. Highly purified CD44+ prostate cancer cells from xenograft human tumors are enriched in tumorigenic and metastatic progenitor cells. Oncogene 2006, 25:1696–708. |
| [74] | Collins AT, Berry PA, Hyde C, Stower MJ, Maitland NJ. Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res 2005, 65:10946–51. |