| Journal of Cancer Stem Cell Research (2013), 1:e1003 © 2013 Creative Commons. All rights reserved ISSN 2329-5872 DOI: 10.14343/JCSCR.2013.1e1003 http://cancerstemcellsresearch.com |
 |
| Journal of Cancer Stem Cell Research (2013), 1:e1003 © 2013 Creative Commons. All rights reserved ISSN 2329-5872 DOI: 10.14343/JCSCR.2013.1e1003 http://cancerstemcellsresearch.com |
 |
| Original Article | Open Access |
| Neonatal exposure to estrogen affects very small ES-like stem cells (VSELs) leading to various pathologies in adults including cancer | |
| Deepa Bhartiya*, Kalpana Sriraman, Smita Bhutda, Akash S. Mundekar, Saim Mulla and Harshada Modak | |
| Stem Cell Biology Department, National Institute for Research in Reproductive Health, Parel, Mumbai 400012, INDIA. | |
| *Correspondence: Tel: +91 22 2419 2012, E-mail: bhartiyad@nirrh.res.in Received: July 20, 2013; Revised: August 10, 2013; Accepted: August 12, 2013 |
Abstract: Very small embryonic-like stem cells (VSELs) are pluripotent stem cells present in various adult body tissues, maintain life-long tissue homeostasis and are possibly the embryonic remnants responsible for cancers. Neonatal exposure to endocrine disruptors is a serious problem and is implicated with several diseases in adults like PCOS in women, infertility in men and increased incidence of endocrine cancers etc. Thus it was of interest to investigate whether VSELs are affected by exposure to endocrine disruptors. Mice pups were injected 20 μg of 17β-estradiol daily on days 5–7 and were sacrificed after 100 days. Testes, ovaries, uteri and prostate tissues were collected and processed for histology and RNA extraction. Testicular sections were also used to immuno-localize PCNA and DAZL, markers that reflect proliferation and germ cell population. Quantitative RT-PCR analysis was performed for Oct-4A and Sca-1, specific markers for VSELs. Histological studies showed PCO-like condition in ovaries, spermatogenesis was affected and both uterus and prostate showed early signs of cancer including hypertrophied and multilayered epithelium, absent glands in the uterus and acute inflammation in prostate. Increased PCNA and DAZL positive cells were detected in the treated testicular sections. Oct-4A and Sca-1 transcripts were detected in all organs and showed increased expression in ovary and testis. Bilateral germ cell tumor was observed in one out of ten treated mice and histological studies showed complete absence of seminiferous tubules and sperm; however VSELs were detected and confirmed by the presence of Oct-4A and Sca-1 transcripts. Present study provides evidence for the first time that neonatal exposure to estrogens results in altered VSELs function in favor of aberrant proliferation rather than differentiation leading to increased risk of cancers. Altered VSELs function possibly due to compromised somatic microenvironment (by neonatal exposure to endocrine disruptors) results in adult onset of various diseases including hormone-sensitive cancers.
Keywords: VSELs, endocrine disruptors, cancer, PCOS, infertility.
Stem cells are considered as the most primitive cells having the ability to self-renew and also differentiate into various cell types in the body. They are broadly classified as (i) adult tissue-specific stem cells and (ii) embryonic stem (ES) cells. ES cells and induced pluripotent stem (iPS) cells are pluripotent in nature (capable to give rise to all the three germ layers in the body) and derived in culture from the inner cell mass of developing blastocyst or by reprogramming somatic cells respectively. There is another group of pluripotent stem cells termed very small embryonic-like stem cells (VSELs) which persist throughout life in adult body tissues, maintain life-long homeostasis (recent review [1]) and are mobilized in case of disease conditions [2–5]. They can be isolated as CD45−Lin−Sca-1+ cells in mice [6] and as CD45−Lin−CD133+ cells in humans [7]. They also express various pluripotent markers including Oct-4A, SSEA-1 (mice), SSEA-4 (humans), Sox-2, Nanog etc. VSELs are relatively quiescent in nature, possibly the primordial germ cells that during their migration along the dorsal mesentery to reach the gonadal ridge during early embryonic development and also get deposited in various body organs [8]. They have been reported in adult mammalian testes and ovary (recent review [9]) and also other adult body organs [10, 11].
It has been recently suggested that various body organs harbor two set of stem cells including very quiescent and actively dividing stem cells [12, 13]. VSELs could possibly be the quiescent stem cell population existing in various adult body tissues and are also proposed to be the embryonic remnants responsible for cancers [14, 15] as proposed 150 years ago by Rudolf Virchow and Julius Conheim. They may be the missing link between embryonic origin of cancer and stem cell theory of cancer development.
One of the serious adverse effects of changing environment due to human interference with nature is the exposure of both humans and animals to endocrine disruptors (EDCs). These are being added into the environment as pesticides, plastics, metal, food cans, toys, cosmetics etc. These toxic substances mimic the endocrine hormones present naturally in the body and depending on the time of exposure lead to developmental, reproductive, neural and immunological problems. Various adverse health effects include (i) malformation of reproductive organs (ii) declining fertility rates (iii) lowered sperm count, early puberty, early reproductive senescence (iv) premature ovarian failure, decreased ovarian reserve, aneuploidy, PCOS (v) increased immunological disorders, (vi) neurodegenerative diseases and most importantly (vii) increase in the incidence of endocrine cancers and other pathologies like endometriosis, fibroids, increased risk of miscarriage etc. [16, 17]. Several indirect linkages of adverse effects of EDCs on endocrine problems are reported in literature. Recently Punjab, the agriculture capital of India (with high exposure to fertilizers, pesticides, DDT etc.) has also been labeled as the cancer capital, possibly due to very high exposure to pesticides and chemicals like arsenic and uranium (http://www.downtoearth.org.in/content/punjab-cancer-capital-india). Various studies to investigate the underlying mechanism of action of the EDCs suggest that DNA methylation pattern i.e. epigenetic changes induced by EDCs result in altered transcription and thus the disease phenotype [18].
Since pluripotent VSELs maintain life-long tissue homoeostasis and are also implicated in cancer initiation, both of which are affected by endocrine disruptors, it was of interest to investigate VSELs status after exposure to endocrine disruptors. Oct-4A and Sca-1 are specific pluripotent markers that reflect the presence of VSELs. It is intriguing to note that common markers exist in literature for pluripotent and cancer stem cells [19]. OCT-4 (POU5F1) acts as a multi-functional factor in both cancer and stem cell biology with a possible oncogenic role as well as a key regulator of self-renewal and differentiation of pluripotent embryonic stem cells [20]. Nuclear Oct-4A isoform is responsible for pluripotency [21, 22] and is also considered as a potential tumor marker [23]. Luo et al [24] recently reported that expression of embryonic stem cells (ESCs) markers including Oct-4 is crucial for progression of various human malignancies.
Besides Oct-4, stem cell antigen-1 (Sca-1 or Ly6A), a cell surface marker has been observed in a variety of tissues like cardiac tissue, mammary glands, skin, muscle, prostate, ovary and testis as well as in bone marrow hematopoietic cells [25]. Sca-1−/− mice have greatly reduced bone mass resulting from a primary defect in the self-renewal capacity of early mesenchymal progenitor cells. In addition, hematopoietic stem cells from Sca-1−/− mice have a decreased repopulation potential. Sca-1+ve cells have increased regenerative capacity compared to Sca-1−ve cells [26]. Also tumors with aggressive phenotype have up regulated Sca-I expression. Thus Sca-1 is important for the self-renewal of stem cells and can be used to enrich stem and progenitor cell populations as well as tumor-initiating cells.
Thus in the present study we have studied by qRT-PCR, the differential expression of Sca-1 and Oct-4A transcripts after peri-natal exposure to pharmacological dose of estrogen (20 μg per day on days 5–7 after birth) in 100 days old adult mice tissues including ovary, testis, uterus and prostate. In addition, we also studied PCNA and DAZL expression in testicular sections which reflect proliferation and differentiation of stem cells into pre-meiotic germ cells.
The study was approved by Institute Animal Ethics Committee and was carried out using in-house bred Swiss mice in the experimental animal facility at our Institute. Mice were housed in a temperature and humidity controlled room on a 12hr light/12hr dark cycle with free access to food and water. 17β-estradiol (Sigma, St. Louis, MO) suspended in sesame oil: alcohol (9:1) was administered subcutaneously to newly born pups daily at a dose of 20 μg/pup on days 5–7. The control group received only the vehicle. The pups were weaned from the mother after a month and were sacrificed when they were 100 days old. Various organs including testis, ovary, uterus and prostate were collected and fixed in neutral buffered formalin for routine histology and also frozen for RNA extraction.
Formalin fixed tissues were processed and embedded in paraffin using standard protocols. Five μm thick sections of the embedded ovaries were prepared and stained with Hematoxylin and Eosin for studying the histo-architecture. The representative areas were photographed using 90i bright field microscope (NIKON, Japan) and data was recorded.
Follicle scoring was done on serial control versus treated ovarian sections to confirm polycystic ovary-like state as reported earlier [27, 28]. Briefly every fifth serial section (8 μm thick) was used for scoring follicles in different stages of growth. The starting section was selected randomly and follicles with clear oocyte nuclei were counted. The cumulative follicle counts were calculated by multiplying with correction factor 5. Randomly 100 testicular tubules were analyzed for stage VIII and IX as described earlier [29] and also for presence of sloughed germ cells in the tubular lumen. In one out of ten male mice, bilateral testicular germ cell tumor was observed at the time of sacrifice. Both the testicles were affected; one was processed for histology and other for RNA studies.
Total RNA was extracted by standard protocol using Trizol (Invitrogen, Carlsbad-CA, USA) and treated with DNase I (Amersham Biosciences, Piscataway, NJ) to remove any genomic DNA as a contaminant. First-strand cDNA was synthesized using the iScript cDNA synthesis kit (Bio-Rad, USA) according to the manufacturer's instructions. Briefly, 1 μg of total RNA was incubated with 5x reaction mix and reverse transcriptase mix. The reaction was carried out in G-STORM thermocycler (Gene Technologies, Braintree, UK). The reaction mix was first incubated at 25°C for 5 min, then at 42°C for 30 min and finally at 85°C for 5 min. qRT-PCR were employed to study gene transcripts expression for Oct-4A (isoform A of octamer binding transcription factor-4; F: CCATGTCCGCCCGCATACGA; R: GGGCTTTCATGTCCTGGGACTCCT) and Sca-1 (stem cell antigen-1; F: AGAGGAAGTTTTATCTGTGCAGCCC and R: TCCACAATAACTGCTGCCTCCTGA).
qRT-PCR was carried out to study the relative expression of Oct-4A and Sca-1 transcripts in treated versus control samples. The expression levels of these gene transcripts in relation to housekeeping gene transcript 18S were estimated by CFX96 Real-time PCR system (Bio-Rad Laboratories, Hercules, CA) using SYBR Green chemistry (Bio-Rad). All the primers had efficiency close to 100%. The amplification conditions included initial denaturation at 94°C for 3 min followed by 40 cycles comprising of denaturation at 94°C for 30 sec, primer annealing (61°C for Oct-4A and 18S, 66°C for Sca-1) for 20 sec and extension at 72°C for 30 sec. The final step included incubation at 94°C for 20 sec to remove any secondary structures followed by melt curve analysis. The fluorescence emitted at each cycle was collected during the extension step of each cycle. The homogeneity of the PCR amplicons was verified by running the products on 2% agarose gels and also by studying the melt curve. All PCR amplifications were carried out in duplicate. Mean Ct values generated in each experiment using the CFX Manager software (Bio-Rad) were used to calculate the mRNA expression levels using ΔΔCt.
Immunolocalization studies were carried out for PCNA and DAZL, to study proliferation status and germ cells in testicular sections. Only testicular sections were used for immunolocalization studies, since stem cells biology is well studied in testes. Briefly, the paraffin sections were deparaffinized, hydrated and antigen retrieval was performed with boiling in SSC buffer, pH 6 in microwave for 15 min. After cooling, the slides were washed with Tris buffer saline (TBS) for 5 min and processed further for blocking with 10% serum (normal horse serum for PCNA and goat serum for DAZL) and 1% BSA in TBS. After removing excess blocking reagent, the slides were incubated with primary antibody diluted with 1% BSA in TBS for two hrs at room temperature (PCNA 1:5000; DAZL 1:100). For negative control, the antibody was omitted and sections were incubated with 1% BSA in TBS. In case of detection alone, negative control sections included were incubated with pre-immune sera from rabbit in which the primary antibody was raised. The detection was done using anti-mouse Vecta ABC kit (Vector Laboratories, USA) according to manufacturer's instructions. Final color development was achieved using DAB (Biogenex, USA). After counterstaining with Haematoxylin, the slides were observed under 90i microscope. Representative areas were photographed.
(a) Ovaries: Ovaries showed basic symptoms of PCO-like condition including presence of follicular cysts and anovulation in agreement with earlier report [30]. Vehicle treated ovary showed presence of follicles in different stages of growth and corpus luteum suggestive of recent ovulation (Figuree 1). In treated ovaries, several follicles were observed; there was abundant stroma and a complete absence of corpus luteum. Cystic follicles were characterized by the presence of fluid filled antrum, thickened thecal layer and atretic changes in the granulosa layer as described earlier [31]. Quantitative analysis of follicles in treated versus control ovaries (n = 2 each) showed an increase in primordial (1545 ± 45 vs. 625 ± 45), primary (240 ± 25 vs. 90 ± 25) and secondary (72 ± 17.5 vs. 17 ± 2.5) follicles however, Graffian follicles and corpus luteum were distinctly absent in treated ovary (Figure 1).
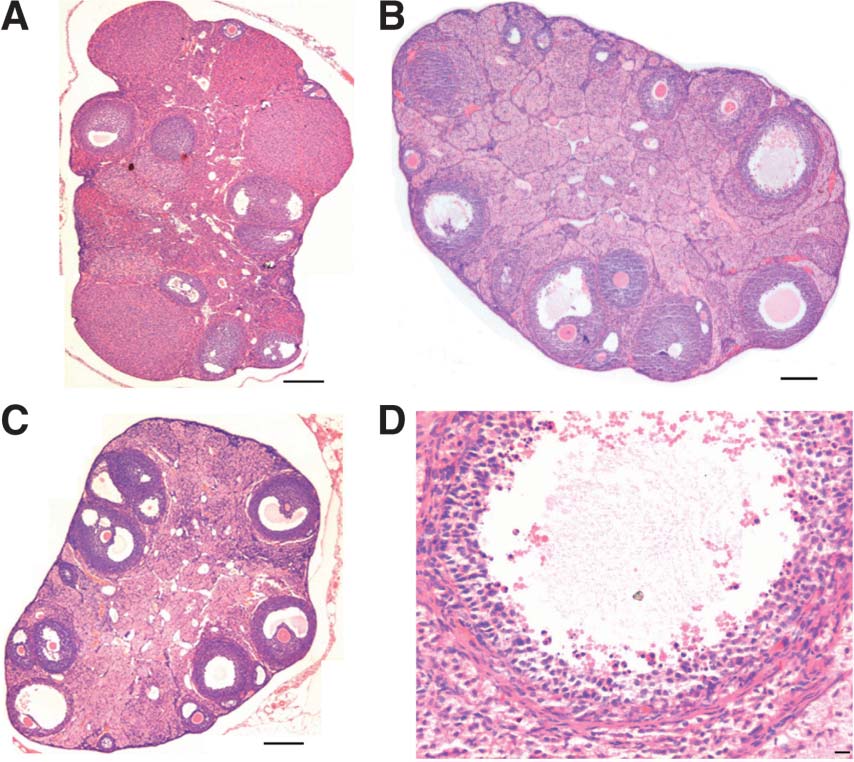 |
Figure 1. Effect of neonatal exposure of estrogen on adult ovaries. Compared to control with several growing follicles and fresh corpus luteum (A) treatment resulted in altered ovarian morphology resembling polycystic ovary-like condition (B & C) This was characterized by absence of corpus luteum, presence of cystic follicles with thickened thecal layer and atretic granulosa cells and abundant stroma. Bar = 100 μm (A–C) and 20 μm (D). |
(b) Testis: Neonatal exposure to estradiol induced subtle changes in testicular spermatogenesis. There was marked sloughing of germ cells in the lumen of seminiferous tubules (Figure 2), in agreement with earlier reports [32]. Interestingly, number of tubules in stages VIII and IX (suggestive of completed spermatogenesis) were counted and were markedly reduced after treatment (Figure 2I). Also number of elongated spermatids per tubule were reduced after treatment (Figure 2G) compared to control (Figure 2D). All these are signs suggestive of compromised fertility.
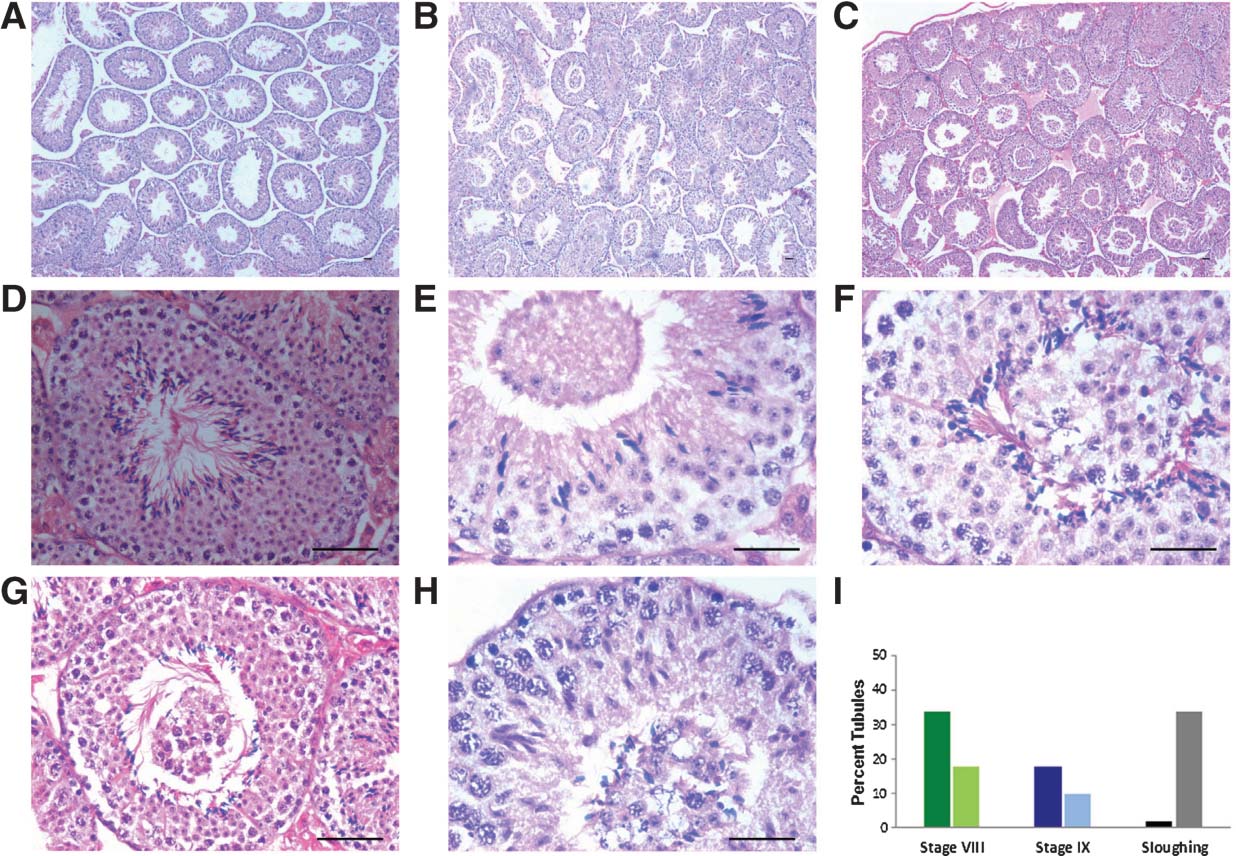 |
Figure 2. Effect of neonatal exposure to estrogen on testicular histology. Normal spermatogenesis is observed in control (A) whereas after treatment marked sloughing of germ cells is observed in the lumen of the seminiferous tubules (B & C). At higher magnification sloughing is clearly evident in the tubular lumen and comprises of undifferentiated germ cells (E–H). Numbers of elongated spermatids are abundant in normal tubule in stage VIII (D) compared to decreased numbers in treated tubule in stage VIII (G). There are signs of compromised differentiation during spermatogenesis. Staging of tubules showed a dramatic reduction in number of tubules at stage VIII and IX and increased sloughing of germ cells after treatment (I). Bar = 20 μm. |
In one mouse, bilateral germ cell tumor (Figure 3) was observed at time of sacrifice. No seminiferous tubules were observed in histological sections (Figure 3) as well as when the other testis was processed for RNA extraction. It was a germ cells tumor and at some places one could observe how the cells may be lost into the peritoneum (Figures 4 & 5). Interestingly these cells were spherical, small in size and with high nucleo-cytoplasmic ratio resembling the VSELs. At places, excessive proliferation of these cells was observed and they also appeared as chains. Higher magnification of the central region of the tumor showed a very different histology (Figure 6). At large number of places, small spherical cells with typical morphology and size of VSELs were observed and appeared as chains, suggestive of rapid proliferation.
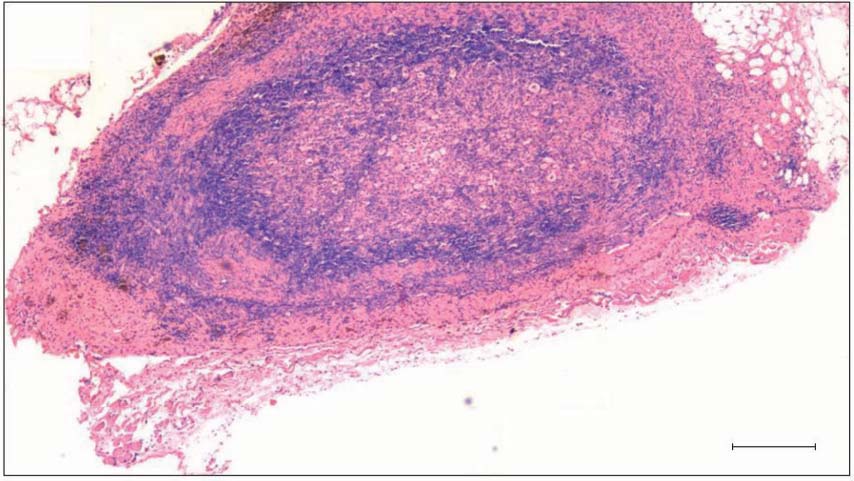 |
Figure 3. Bilateral testicular tumor was observed in 1 out of 10 treated mice at the time of sacrifice. Cross-section of testis showed absence of seminiferous tubules and sperm. Bar = 100 μm. |
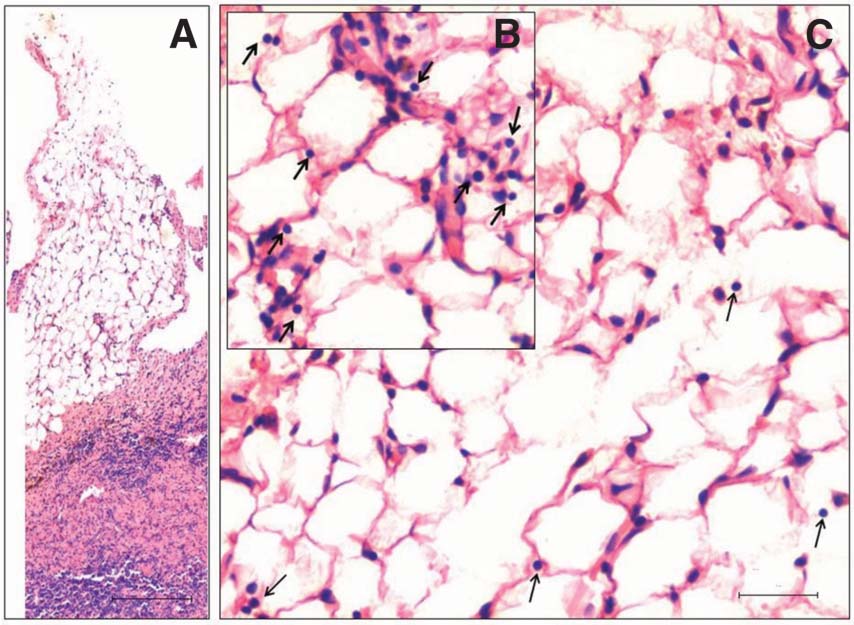 |
Figure 4. At places the testicular membrane was expanded into the peritoneum and possibly shows how metastasis occurs (A). At higher magnification, small spherical cells with high nucleo-cytoplasmic ratio suggestive of VSELs were observed (arrows, B & C). This plate shows how VSELs possibly escape from the primary tumor site and metastasize to other organs. Bar = 100 μm (A) and 20 μm (C). |
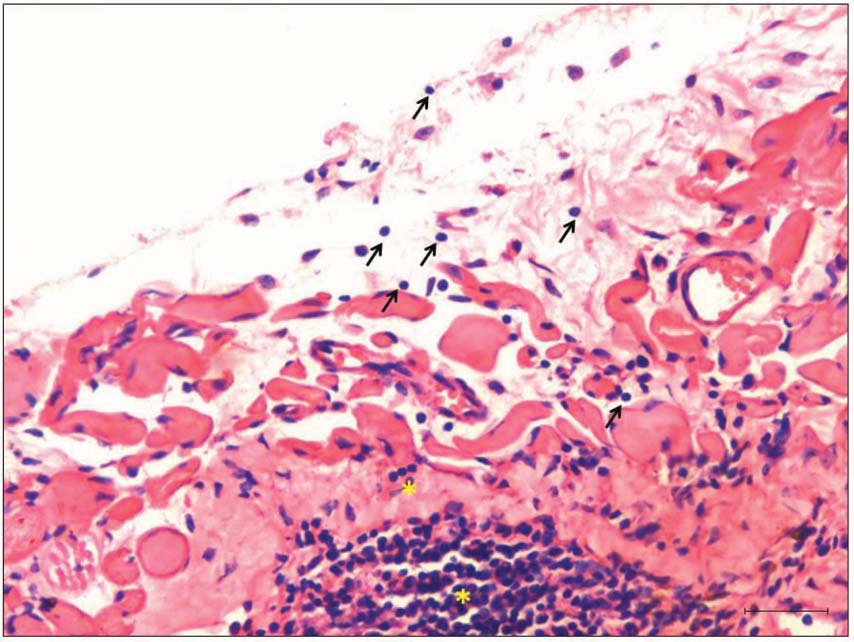 |
Figure 5. Another field on testicular surface where VSELs were observed escaping (arrow) into the peritoneum. The VSELs were present in large numbers and also as chains reflecting extensive proliferation (yellow asterisks). This abnormal proliferation of VSELs is observed in tumor tissue whereas in normal tissues these cells are relatively quiescent whereas the immediate descendants ‘progenitors’ expand rapidly to maintain homeostasis [45]. Bar = 20 possibly. |
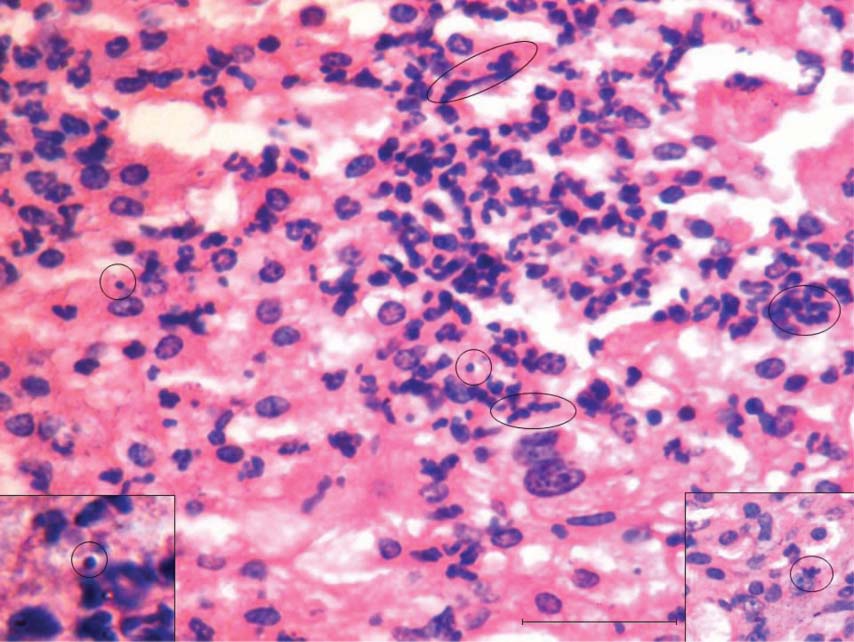 |
Figure 6. In the center of the testicular tissue shown in Fig 3, please note extensive proliferation of very small spherical stem cells which are presumably the VSELs. Circled areas represent extensive proliferation of VSELs. The tumor appears to have originated here and later the VSELs escape to the surface and the peritoneum (Figures 3 and 4). Bar = 20 μm. |
(c) Uterus: Uterine histology was grossly affected by the treatment (Figure 7). Normal uterus showed abundant glands (Figure 7A and C) compared to a dramatic absence of glands after treatment (Figure 7B and D). The surface epithelium lining the lumen was multilayered and hypertrophied after treatment (Figure 7E–G) compared to control (Figure 7C). Uterine gland development termed adenogenesis occurs during neonatal stage from the luminal epithelium [33, 34] and appears to be under the control of estradiol and Wnt7A [35] and absent glands will result in implantation failure [36].
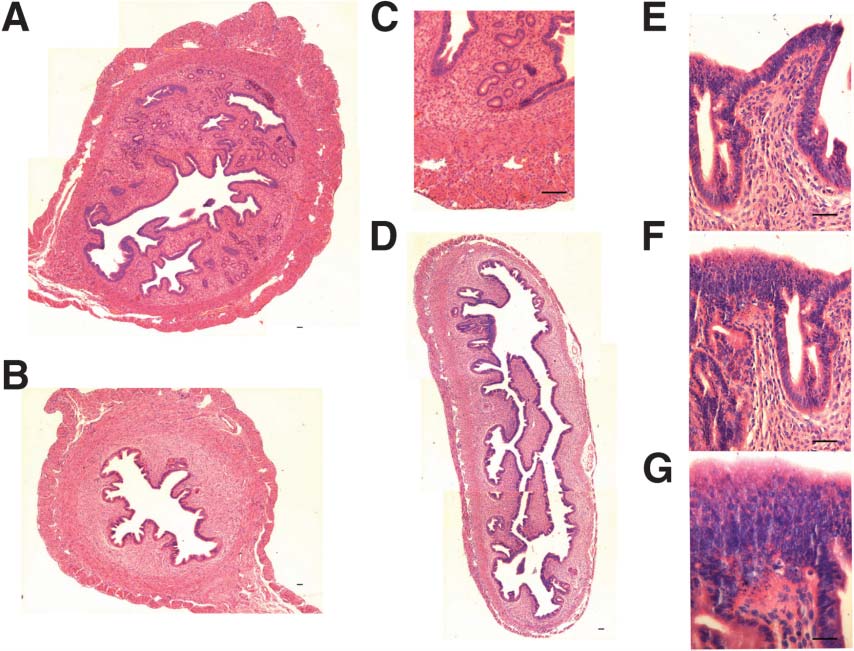 |
Figure 7. Effect of neonatal exposure of estrogen on mouse uterine histology. Normal uterus (A & C) showed presence of abundant glands and epithelial cells lining the luminal and glandular epithelium. Treatment affects differentiation of uterine glands from the luminal epithelium are dramatically lacking after treatment (B & D). At higher magnification the epithelial cells lining the uterine lumen are hypertrophied and multilayered indicating increased risk of cancer (E–G). Bar = 20 μm. |
(d) Prostate: Characteristic signs of epithelial hyperplasia and dysplasia were evident after treatment (Figure 8C–F) compared to control prostate (Figure 8A–B). In addition inflammatory response was also noted suggestive of early signs of cancer in agreement with earlier reports [37, 38]. Inflammation in prostate has been linked to the pathogenesis of cancers [39]. Estrogens are essential for normal homeostasis within the prostate and any change leads to perturbation of the glands growth and the emergence of disease.
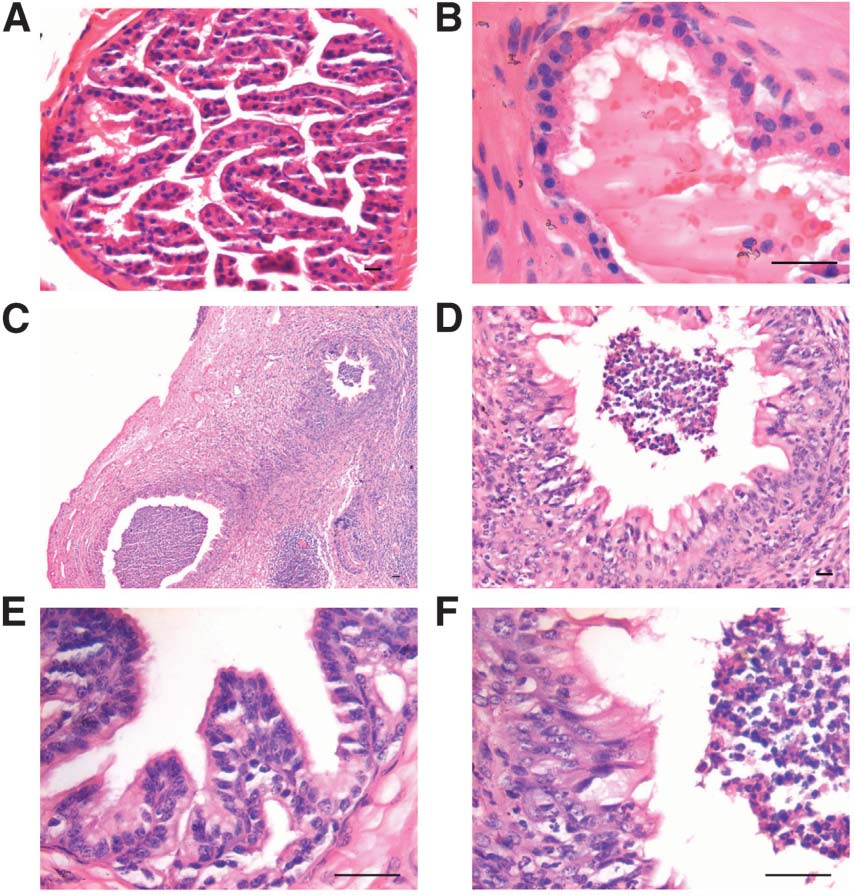 |
Figure 8. Effect of neonatal exposure of estrogen on the prostrate. Compared to control prostate section (A–B), neonatal exposure to estrogen led to massive inflammatory response (C–D & E) and hypertrophied epithelium. The inflammatory cells in the stroma migrated through the epithelium into the lumen also (D & F). Bar = 20 μm. |
Both control and treated organs showed the presence of Oct-4A and Sca-1 transcripts of expected size (Figure 9). Studies performed using RNA extracted from tissues collected from 2 control and 2 treated mice and repeated twice showed a two-fold increased expression of VSELs specific markers Oct-4A and Sca-1 in the ovarian and testicular tissues. However, a clear picture did not emerge for prostate and uterus. In prostate possibly because of excessive inflammation, qRT-PCR studies may not be the right method to study the effect of treatment on VSELs. We recently reported the presence of VSELs in mouse uterus and that they may be involved in the regular repair and regeneration [40]. Time kinetic studies may help for a clearer picture to emerge about the possible role of VSELs in treated uterus and prostate resulting in various pathologies, including cancer. Interestingly, RT-PCR studies showed the presence of both Oct-4A and Sca-1 in the mouse testicular tumor sample (data not shown).
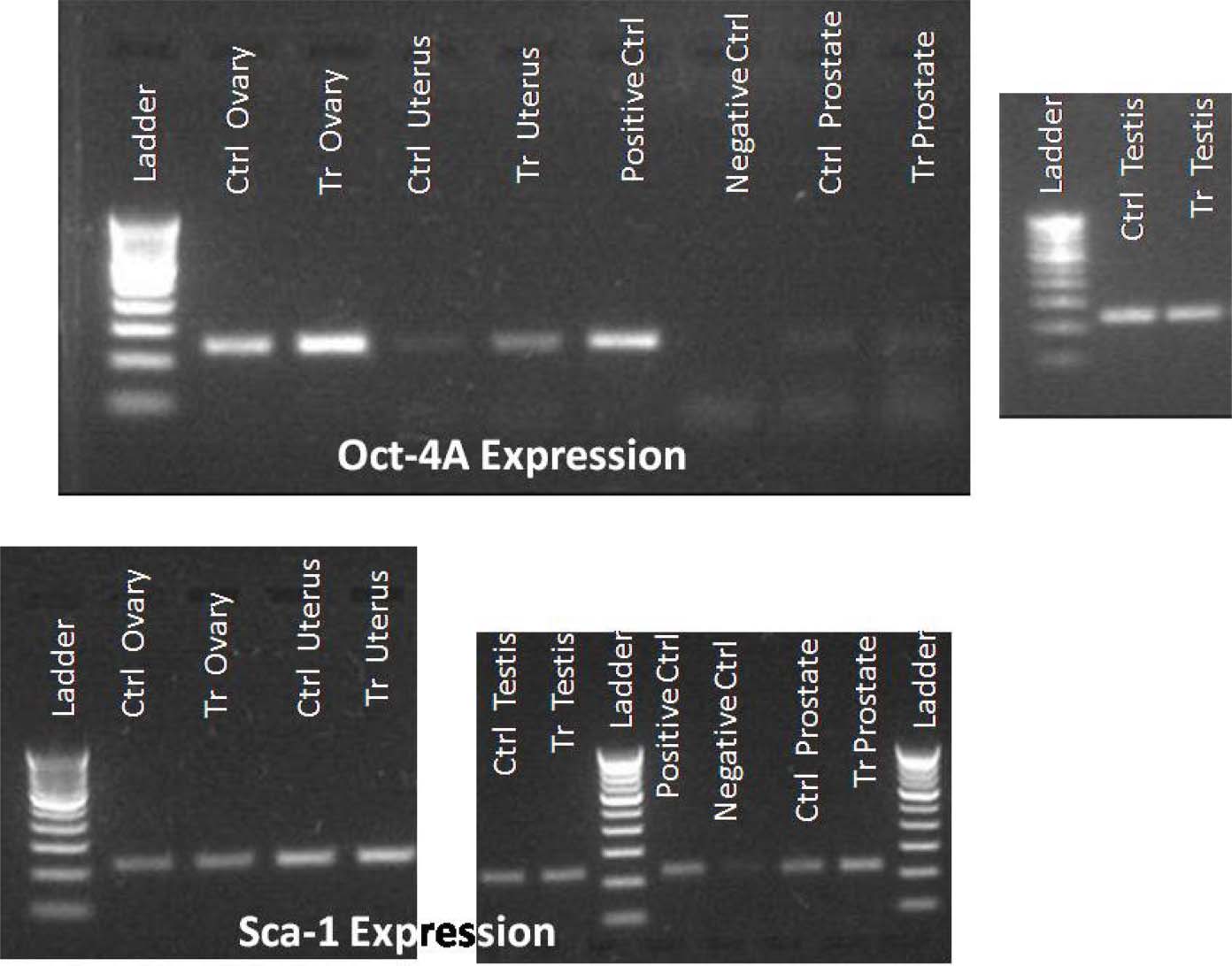 |
Figure 9. Analysis of VSELs markers Oct-4A and Sca-1 by RT-PCR. Both control and treated organs show presence of transcripts for Oct-4A and Sca-1. The positive control used is normal ovarian sample and negative control is no template control of PCR. 100 base pair ladder is used. |
PCNA was immuno-localized on normal testicular sections and few cells towards the basement membrane stained positive (Figure 10A–C). The staining was observed in a few random tubules whereas in the treated group, all the mitotically proliferating spermatogonia present in 1–3 layers stained positive (Figure 10D–F). Interestingly the size of cells stained positive for PCNA also showed a variation in size which was clearly observed at higher magnification (Figure 10F). The smaller cells are possibly the spermatogonial stem cells and showed nuclear PCNA whereas the bigger spermatogonia or spermatocytes showed cytoplasmic localization of PCNA which eventually disappeared as germ cells underwent meiosis and differentiated further. DAZL, a premeiotic germ cell marker was also detected in abundance after treatment (Figure 11C–F) compared to control (Figure 11A–B).
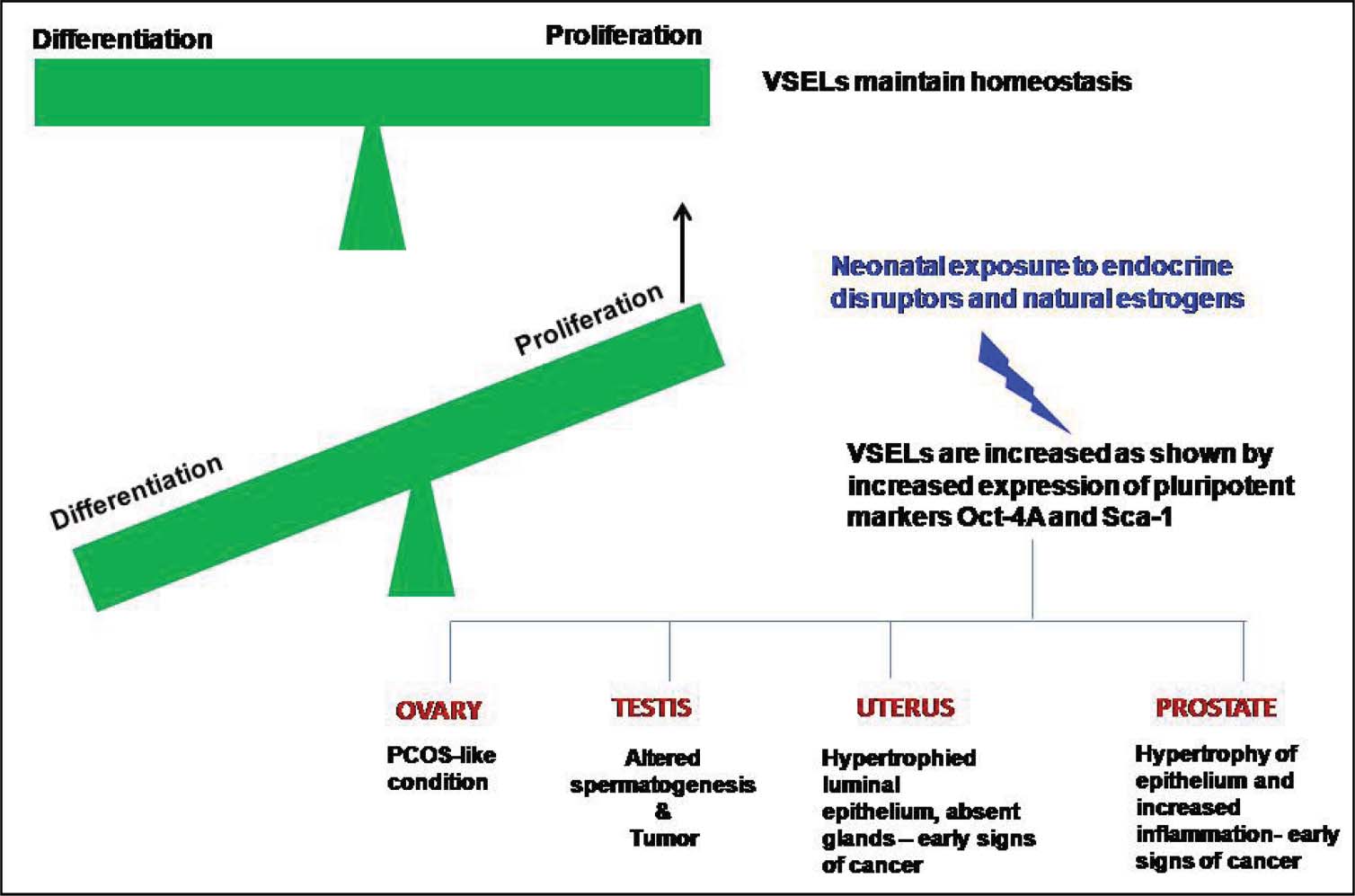 |
Figure 10. Immunolocalization of proliferating cell nuclear antigen (PCNA) on testicular sections in control (A–C) and treated (D–F) groups. In control sections, PCNA is strictly nuclear and present in few scattered cells at the basement membrane of few testicular tubules, which are probably the spermatogonial stem cells. Treatment resulted in increased PCNA staining. PCNA was localized in 1–3 layers in all the cells lining the basement membrane of all the tubules. PCNA staining was clearly dark and nuclear in small cells and was also observed in bigger cells where it varied from nuclear to cytoplasmic. Bar = 20 μm. |
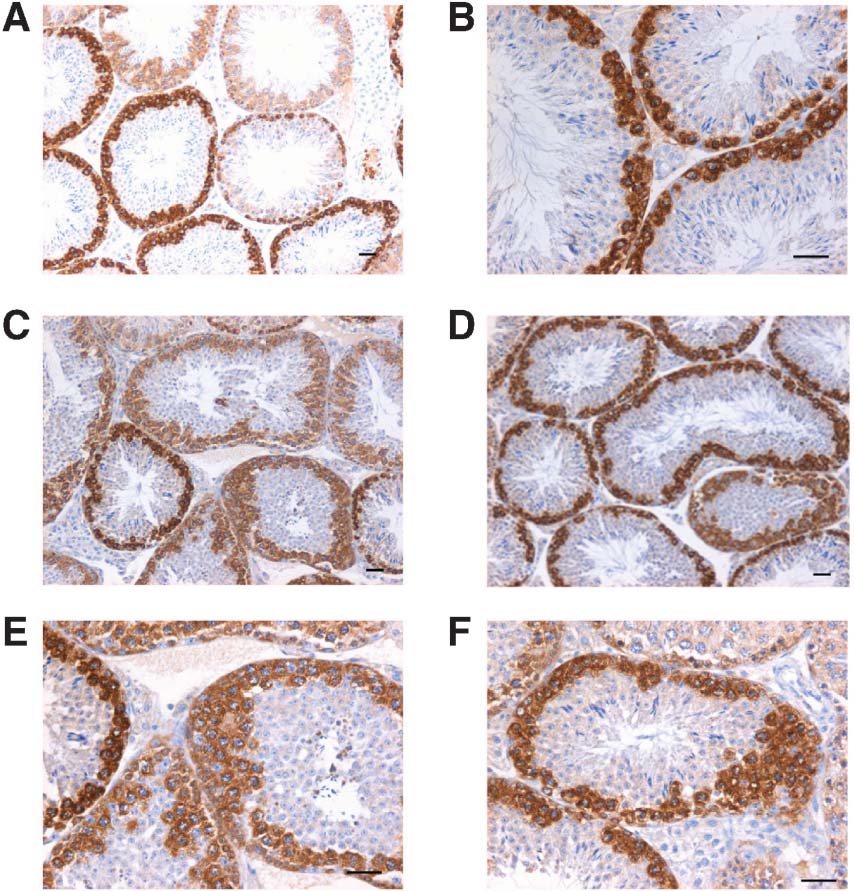 |
Figure 11. Immunolocalization of DAZL (pre-meiotic marker) in control (A, B) and treated testicular sections (C–F). An increased DAZL expression was clearly evident after treatment suggestive of increased proliferation of pre-meiotic germ cells. Bar = 20 μm. |
Neonatal exposure to estradiol induced gross changes in the gonads, uterus as well as in the prostate, in agreement with available published reports. In the available literature, these changes have been co-related with altered epigenetic changes [41]; inability of the thymus to produce regulatory T cells resulting in PCOS [30] etc. However, the present study shows a possible link of these disease phenotypes including cancers with VSELs, in agreement with the published hypothesis [14, 15]. Thus altered epigenetic changes reported in available literature could be explained to occur due to compromised differentiation of VSELs.
VSELs are the most primitive embryonic-like stem cells present in adult body organs and sit at the top in the hierarchy in bone marrow stem cells, MSCs and also in other body organs including ovary and testes [9, 42]. Taichman's group [43] recently implicated LIN-/CD45-/SCA-1+ stem cells (VSELs) in prostate metastasis. Ratajczak's group has recently reported higher numbers of circulating VSELs and MSCs in pancreatic cancer patients [44]. Bilateral testicular tumor observed in 1/10 treated mice in the present study, provides direct evidence that VSELs are the embryonic remnants in adult body tissues that may be implicated in testicular cancers. In normal testis, generally cells slightly bigger than the VSELs ‘progenitors’ show clonal expansion whereas the VSELs are rare and invariably detected singly due to their quiescent state [45]. Thus, it is possible that rapid proliferation of VSELs (rather than rare asymmetric cell division under normal conditions) as observed in the present study (Figures 5 and 6) results in tumor formation. Evidently the tumor was induced because of the altered testicular niche due to neonatal exposure to estradiol. It is becoming evident that aggressive tumors express pluripotent markers as reported by several independent groups in various kinds of tumor [19, 20, 23, 24, 46–49]. What could be the cell source expressing pluripotent markers? It is likely that pluripotent VSELs could possibly be the cancer stem cells (CSCs), however what triggers this change of normal functional VSELs in to CSCs is presently not understood.
Few groups have co-related conditions like PCOS with stem cells [50]. Similarly Hu et al [51] have also proposed that endocrine disruptors may act on prostate stem/progenitor cells resulting in increased risk of cancers. Leong et al [52] reported the generation of prostate acini from a single stem cell which was hormone-insensitive and Lin(-)Sca-1(+)CD133(+)CD44(+)CD117(+). We believe that these cells with amazing regenerative ability are probably the VSELs and warrants further investigation. VSELs are emerging as strong candidates that maintain normal homeostasis in adult body organs, are mobilized in disease conditions to bring about regeneration and also may be responsible for various cancers under aberrant conditions.
Results of the present study suggest that various diseases like PCO, infertility and cancer occurred due to a tilt of stem cell function towards proliferation rather than differentiation (Figure 7). This happened because of an altered somatic microenvironment. Generally the VSELs remain quiescent and the progenitors divide rapidly and differentiate resulting in tissue homeostasis. Stem cell interaction with its niche is crucial for their proper function and to maintain a balance between proliferation and differentiation. Niche comprises of somatic cells that support the stem cell and are a source of growth factors and cytokines [53]. It has also been proposed that a compromised microenvironment leads to menopause (cessation of stem cell differentiation into new follicles) [54]. We detected ovarian stem cells (VSELs) in a sixty year old women ovary which differentiated into oocyte-like structures in culture but were not functional in situ because of a compromised niche (personal observations). Several recent reviews have correlated changes in microenvironment to tumor development [55]. Normal cellular microenvironment keeps the stem cells under control and can inhibit malignant cell growth; and changes occurring in tumor microenvironment synergistically support uncontrolled stem cell proliferation. This was clearly observed in the present study (Figures 3–6). In the testicular tumor, VSELs appeared to expand in large numbers. However, the nature of the molecular alterations underlying these changes in the somatic microenvironment still remains unclear. The reciprocal interaction of epithelium and underlying mesenchyme is crucial for normal development and also for carcinogenesis [56]. This interaction appeared to be perturbed in both the uterus and prostate. As a result there was complete absence of glands in treated uterus and marked inflammation in the prostate. Both the organs exhibited hypertrophied, multilayered epithelium and early signs of cancer.
When a pluripotent stem cell becomes committed extensive epigenetic changes occur, the euchromatin in the pluripotent stem cells gets compacted and becomes inactive. Only the genes specific for the fate to which the stem cell gets committed remain active. Somehow the differentiation ability of the VSELs is compromised in the affected organs resulting in manifestation of disease in adult life due to fetal exposure to endocrine disruptors. It would be of interest to discuss here that epigenetic changes reported in the past by various investigators [41] or the inability of thymus to produce T cells [30] due to exposure to endocrine disruptors is a cause or effect of the treatment. We propose that all these changes are indeed the altered effects due to impaired differentiation of VSELs.
Present study is like hitting the tip of an iceberg and provides newer avenues for research and better understanding of adverse effects induced by the endocrine disruptors. There is compelling evidence that altered epithelial stromal interaction leads to altered microenvironment that tilts VSELs function more towards proliferation leading to various diseases (Figure 12). To conclude, in contrast to the recent news where Weissman's group has questioned the very presence of VSELs in adult mice tissues [57], present study provides evidence that VSELs exist in adult tissues and are indeed the embryonic remnants responsible for increased incidence of cancers that arise due to compromised niche (unable to keep a check on VSELs function) with advanced age. Million dollar question remains that what could be done to reverse this tilt back to normal.
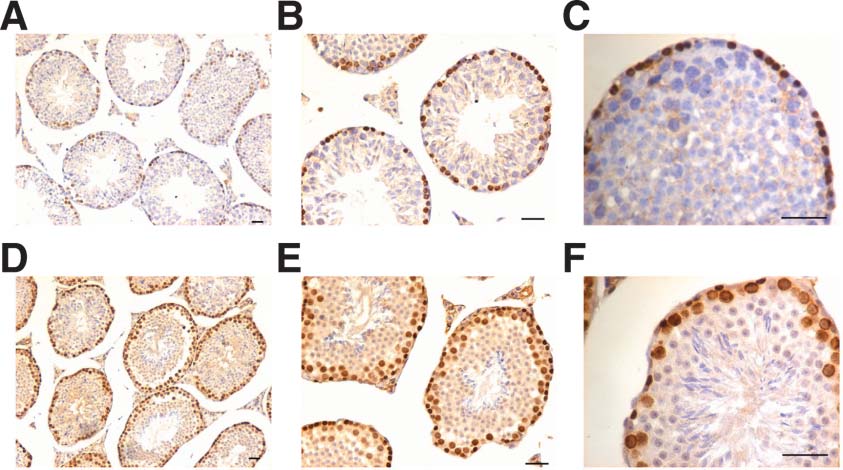 |
Figure 12. Proposed model explaining a central mechanism that is possibly affected by neo-natal exposure to estrogens (endocrine disruptors). Increased number of VSELs and their tilt in favor of proliferation rather than differentiation may be responsible for adult manifestation of various diseases including cancer. |
Conflict of interest: Authors declare no conflict of interest.
We acknowledge the work of several of our colleagues working world-wide on endocrine disruptors and related fields, whose work is very relevant but has not been cited. This study was supported by NIRRH core support (Indian Council of Medical Research, Government of India, New Delhi). We also acknowledge the efforts put in by graduate students Sakina and Rupal, and help of Dr. Rohit Dhumal, Toxicology Dept and Dr. Nafisa Balasinor, Neuroendocrinology Dept, NIRRH for expert help in prostate pathology and staging of testicular tubules respectively.
| [1] | Ratajczak, MZ. Zuba-Surma, E. Kucia, M. Poniewierska, A. Suszynska, M. Ratajczak, J. Pluripotent and multipotent stem cells in adult tissues. Adv Med Sci 2012, 57:1–17. |
| [2] | Borlongan, CV. Glover, LE. Tajiri, N. Kaneko, Y. Freeman, TB. The great migration of bone marrow-derived stem cells toward the ischemic brain: therapeutic implications for stroke and other neurological disorders. Prog Neurobiol 2011, 95:213–28. |
| [3] | Drukała, J. Paczkowska, E. Kucia, M. Młyńska, E. Krajewski, A. Machaliński, B. Madeja, Z. Ratajczak, MZ. Stem cells, including a population of very small embryonic-like stem cells, are mobilized into peripheral blood in patients after skin burn injury. Stem Cell Rev 2012, 8:184–94. |
| [4] | Wojakowski, W. Kucia, M. Liu, R. Zuba-Surma, E. Jadczyk, T. Bachowski, R. Nabiałek, E. Kaźmierski, M. Ratajczak, MZ. Tendera, M. Circulating very small embryonic-like stem cells in cardiovascular disease. J Cardiovasc Transl Res 2011, 4:138–44. |
| [5] | Wojakowski, W. Ratajczak, MZ. Tendera, M. Mobilization of very small embryonic-like stem cells in acute coronary syndromes and stroke. Herz. 2010, 35:467–72. |
| [6] | Kucia, M. Reca, R. Campbell, FR. Zuba-Surma, E. Majka, M. Ratajczak, J. Ratajczak, MZ. A population of very small embryonic-like (VSEL) CXCR4 (+) SSEA-1(+) Oct-4+ stem cells identified in adult bone marrow. Leukemia 2006, 20:857–69. |
| [7] | Kucia, M. Halasa, M. Wysoczynski, M. Baskiewicz-Masiuk, M. Moldenhawer, S. Zuba-Surma, E. Czajka, R. Wojakowski, W. Machalinski, B. Ratajczak, MZ. Morphological and molecular characterization of novel population of CXCR4+ SSEA-4+ Oct-4+ very small embryonic-like cells purified from human cord blood: preliminary report. Leukemia 2007, 21:297–303. |
| [8] | Kucia, M. Machalinski, B. Ratajczak, MZ. The developmental deposition of epiblast/germ cell-line derived cells in various organs as a hypothetical explanation of stem cell plasticity?Acta Neurobiol Exp (Wars) 2006, 66:331–41. |
| [9] | Bhartiya, D. Unni, S. Parte, S. Anand, S. Very small embryonic-like stem cells: Implications in reproductive biology. Biomed Res Int 2013, 682326. |
| [10] | Zuba-Surma, EK. Kucia, M. Ratajczak, J. Ratajczak, MZ. “Small stem cells” in adult tissues: very small embryonic-like stem cells stand up!Cytometry A 2009, 75:4–13. |
| [11] | Ratajczak, MZ. Kucia, M. Ratajczak, J. Zuba-Surma, EK. A multi-instrumental approach to identify and purify very small embryonic like stem cells (VSELs) from adult tissues. Micron 2009, 40:386–93. |
| [12] | Li, L. Clevers, H. Coexistence of quiescent and active adult stem cells in mammals. Science 2010, 327:542–5. |
| [13] | De Rosa, L. De Luca, M. Cell biology: Dormant and restless skin stem cells. Nature 2012, 489:215–17. |
| [14] | Ratajczak, MZ. Shin, DM. Kucia, M. Very small embryonic/epiblast-like stem cells: A missing link to support the germ line hypothesis of cancer development?Am J Pathol. 2009, 174:1985–92. |
| [15] | Ratajczak, MZ. Shin, DM. Liu, R. Marlicz, W. Tarnowski, M. Ratajczak, J. Kucia, M. Epiblast/germ line hypothesis of cancer development revisited: lesson from the presence of Oct-4+ cells in adult tissues. Stem Cell Rev 2010, 6:307–16. |
| [16] | Schug, TT. Janesick, A. Blumberg, B. Heindel, JJ. Endocrine disrupting chemicals and disease susceptibility. J Steroid BiochemMolBiol 2011, 127:204–15. |
| [17] | Soto, AM. Sonnenschein, C. Environmental causes of cancer: Endocrine disruptors as carcinogens. Nat Rev Endocrinol 2010, 6:363–70. |
| [18] | Anway, MD. Skinner, MK. Epigenetic transgenerational actions of endocrine disruptors. Endocrinology 2006, 147:S43–9. |
| [19] | Monk, M. Holding, C. Human embryonic genes re-expressed in cancer cells. Oncogene 2001, 20:8085–91. |
| [20] | Gidekel, S. Pizov, G. Bergman, Y. Pikarsky, E. Oct-3/4 is a dose-dependent oncogenic fate determinant. Cancer Cell 2003, 4:361–70. |
| [21] | Liedtke, S. Enczmann, J. Waclawczyk, S. Wernet, P. Kögler, G. Oct4 and its pseudogenes confuse stem cell research. Cell Stem Cell 2007, 1:364–66. |
| [22] | Wang, X. Dai, J. Concise review: isoforms of OCT4 contribute to the confusing diversity in stem cell biology. Stem Cells 2010, 28:885–93. |
| [23] | Atlasi, Y. Mowla, SJ. Ziaee, SA. Bahrami, AR. OCT-4, an embryonic stem cell marker, is highly expressed in bladder cancer. Int J Cancer 2007, 120:1598–602. |
| [24] | Luo, W. Li, S. Peng, B. Ye, Y. Deng, X. Yao, K. Embryonic stem cells markers SOX2, OCT4 and Nanog expression and their correlations with epithelial-mesenchymal transition in nasopharyngeal carcinoma. PLoS One 2013, 8:e56324. |
| [25] | Holmes, C. Stanford, WL. Concise review: stem cell antigen-1: expression, function, and enigma. Stem Cells 2007, 25:1339–47. |
| [26] | Ito, CY. Li, CY. Bernstein, A. Dick, JE. Stanford, WL. Hematopoietic stem cell and progenitor defects in Sca-1/Ly-6A-null mice. Blood. 2003, 101:517–23. |
| [27] | Tilly, JL. Ovarian follicle counts—not as simple as 1, 2, 3. Reprod Biol Endocrinol 2003, 1:11. |
| [28] | Maciel, GA. Baracat, EC. Benda, JA. Markham, SM. Hensinger, K. Chang, RJ. Erickson, GF. Stockpiling of transitional and classic primary follicles in ovaries of women with polycystic ovary syndrome. J Clin Endocrinol Metab 2004, 89:5321–7. |
| [29] | Hess, RA. Renato de Franca, L. Spermatogenesis and cycle of the seminiferous epithelium. Adv Exp Med Biol 2008, 636:1–15. |
| [30] | Chapman, JC. Min, SH. Freeh, SM. Michael, SD. The estrogen-injected female mouse: new insight into the etiology of PCOS. Reprod Biol Endocrinol. 2009, 7:47. |
| [31] | Maharjan, R. Nagar, PS. Nampoothiri, L. Effect of Aloe barbadensis Mill. formulation on Letrozole induced polycystic ovarian syndrome rat model. J Ayurveda Integr Med 2010, 1:273–9. |
| [32] | Salian, S. Doshi, T. Vanage, G. Perinatal exposure of rats to Bisphenol A affects the fertility of male offspring. Life Sci 2009, 85:742–52. |
| [33] | Carpenter, KD. Gray, CA. Bryan, TM. Welsh, TH. Spencer, TE. Estrogen and antiestrogen effects on neonatal ovine uterine development. Biol Reprod. 2003, 69:708–17. |
| [34] | Gray, CA. Bartol, FF. Tarleton, BJ. Wiley, AA. Johnson, GA. Bazer, FW. Spencer, TE. Developmental biology of uterine glands. Biol Reprod. 2001, 65:1311–23. |
| [35] | Dunlap, KA. Filant, J. Hayashi, K. Rucker, EB. Song, G. Deng, JM. Behringer, RR. DeMayo, FJ. Lydon, J. Jeong, JW. Spencer, TE. Postnatal deletion of Wnt7a inhibits uterine gland morphogenesis and compromises adult fertility in mice. Biol Reprod 2011, 85:386–96. |
| [36] | Filant, J. Spencer, TE. Endometrial glands are essential for blastocyst implantation and decidualization in the mouse uterus. Biol Reprod 2013, 88:93. |
| [37] | Bianco, JJ. Handelsman, DJ. Pedersen, JS. Risbridger, GP. Direct response of the murine prostate gland and seminal vesicles to estradiol. Endocrinology 2002, 143:4922–33. |
| [38] | Bianco, JJ. McPherson, SJ. Wang, H. Prins, GS. Risbridger, GP. Transient neonatal estrogen exposure to estrogen-deficient mice (aromatase knockout) reduces prostate weight and induces inflammation in late life. Am J Pathol 2006, 168:1869–78. |
| [39] | De Marzo, AM. Platz, EA. Sutcliffe, S. Xu, J. Grönberg, H. Drake, CG. Nakai, Y. Isaacs, WB. Nelson, WG. Inflammation in prostate carcinogenesis. Nat Rev Cancer 2007, 7:256–69. |
| [40] | Gunjal, P. Bhartiya, D. Metkari, S. Manjramkar, D. Are very small embryonic stem cells (VSELs) the elusive endometrial stem cells responsible for regeneration? Reproduction (Manuscript under review). |
| [41] | Zhang, X. Ho, SM. Epigenetics meets endocrinology. J Mol Endocrinol 2011, 46:R11–32. |
| [42] | Havens, AM. Shiozawa, Y. Jung, Y. Sun, H. Wang, J. McGee, S. Mishra, A. Taichman, LS. Danciu, T. Jiang, Y. Yavanian, G. Leary, E. Krebsbach, PH. Rodgerson, D. Taichman, RS. Human very small embryonic-like cells generate skeletal structures, in vivo. Stem Cells Dev 2013, 22:622–30. |
| [43] | Jung, Y. Kim, JK. Shiozawa, Y. Wang, J. Mishra, A. Joseph, J. Berry, JE. McGee, S. Lee, E. Sun, H. Wang, J. Jin, T. Zhang, H. Dai, J. Krebsbach, PH. Keller, ET. Pienta, KJ. Taichman, RS. Recruitment of mesenchymal stem cells into prostate tumours promotes metastasis. Nat Commun 2013, 4:1795. |
| [44] | Starzyńska, T. Dąbkowski, K. Błogowski, W. Zuba-Surma, E. Budkowska, M. Sałata, D. Dołęgowska, B. Marlicz, W. Lubikowski, J. Ratajczak, MZ. An intensified systemic trafficking of bone marrow-derived stem/progenitor cells in patients with pancreatic cancer. J Cell Mol Med 2013, 17(6):792–9. |
| [45] | Bhartiya, D. Kasiviswanathan, S. Unni, SK. Pethe, P. Dhabalia, JV. Patwardhan, S. Tongaonkar, HB. Newer insights into premeiotic development of germ cells in adult human testis using Oct-4 as a stem cell marker. J Histochem Cytochem 2010, 58:1093–106. |
| [46] | Ling, GQ. Chen, DB. Wang, BQ. Zhang, LS. Expression of the pluripotency markers Oct3/4, Nanog and Sox2 in human breast cancer cell lines. Oncol Lett 2012, 4:1264–8. |
| [47] | Guo, Y. Liu, S. Wang, P. Zhao, S. Wang, F. Bing, L. Zhang, Y. Ling, EA. Gao, J. Hao, A. Expression profile of embryonic stem cell-associated genes OCT-4, Sox2 and Nanog in human gliomas. Histopathology 2011, 59:763–75. |
| [48] | Wang, X. Ouyang, H. Yamamoto, Y. Kumar, PA. Wei, TS. Dagher, R. Vincent, M. Lu, X. Bellizzi, AM. Ho, KY. Crum, CP. Xian, W. McKeon, F. Residual embryonic cells as precursors of a Barrett's-like metaplasia. Cell 2011, 145:1023–35. |
| [49] | Schoenhals, M. Kassambara, A. De Vos, J. Hose, D. Moreaux, J. Klein, B. Embryonic stem cell markers expression in cancers. Biochem Biophys Res Commun 2009, 383:157–62. |
| [50] | Tilly, JL. Rueda, BR. Mini review: stem cell contribution to ovarian development, function, and disease. Endocrinology 2008, 149:4307–11. |
| [51] | Hu, WY. Shi, GB. Hu, DP. Nelles, JL. Prins, GS. Actions of estrogens and endocrine disrupting chemicals on human prostate stem/progenitor cells and prostate cancer risk. Mol Cell Endocrinol 2012, 354:63–73. |
| [52] | Leong, KG. Wang, BE. Johnson, L. Gao, WQ. Generation of a prostate from a single adult stem cell. Nature 2008, 456:804–08. |
| [53] | Scadden, DT. The stem-cell niche as an entity of action. Nature 2006, 441:1075–79. |
| [54] | Massasa, E. Costa, XS. Taylor, HS. Failure of the stem cell niche rather than loss of oocyte stem cells in the aging ovary. Aging 2010, 2:1–2. |
| [55] | Honoki, K. Fujii, H. Tsujiuchi, T. Cancer Stem Cell Niche: The role of mesenchymal stem cells in tumor microenvironment. In Cancer Stem Cells –, The Cutting Edge, Ed. Prof. Shostak S. Chapter 10: 2011, 189–206. |
| [56] | Ingber, DE. Cancer as a disease of epithelial-mesenchymal interactions and extracellular matrix regulation. Differentiation 2002, 70:547–60. |
| [57] | Abbott, A. Doubt cast over tiny stem cells. Nature 2013, 499:390. |